Seznamy Atom Model Of Magnesium
Seznamy Atom Model Of Magnesium. This means it contains 12 protons, 12 neutrons and 12 electrons. It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element. 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305.
Nejchladnější Chemical Elements Com Magnesium Mg
At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305. 12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons.This means it contains 12 protons, 12 neutrons and 12 electrons.
Its key isotope is mg25 and there is also a rare isotope of mg26. Belonging to group 2 & … Its key isotope is mg25 and there is also a rare isotope of mg26. At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element.

Belonging to group 2 & … Its key isotope is mg25 and there is also a rare isotope of mg26... With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu.

12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons. 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305. It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element. Belonging to group 2 & … This means it contains 12 protons, 12 neutrons and 12 electrons. It contains three electron shells, of which the first contains two … Its key isotope is mg25 and there is also a rare isotope of mg26. With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu. At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. 12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons.. At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c.

12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305... It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element. 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305. At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. Its key isotope is mg25 and there is also a rare isotope of mg26. With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu.

With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu. At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. Belonging to group 2 & … This means it contains 12 protons, 12 neutrons and 12 electrons. It contains three electron shells, of which the first contains two … It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element. 12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons. Its key isotope is mg25 and there is also a rare isotope of mg26. 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305. With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu.. Its key isotope is mg25 and there is also a rare isotope of mg26.

12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305... At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. This means it contains 12 protons, 12 neutrons and 12 electrons. 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305. With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu... With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu.

This means it contains 12 protons, 12 neutrons and 12 electrons... It contains three electron shells, of which the first contains two … 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305.. This means it contains 12 protons, 12 neutrons and 12 electrons.

It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element. At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu. 12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons. It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element. This means it contains 12 protons, 12 neutrons and 12 electrons. 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305. It contains three electron shells, of which the first contains two … Belonging to group 2 & … Its key isotope is mg25 and there is also a rare isotope of mg26.. At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c.
/GettyImages-1135707671-640473b29d534e15a24491c0d6b2789e.jpg)
Belonging to group 2 & … It contains three electron shells, of which the first contains two … 12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons. It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element. This means it contains 12 protons, 12 neutrons and 12 electrons. At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c.

It contains three electron shells, of which the first contains two …. .. This means it contains 12 protons, 12 neutrons and 12 electrons.
It contains three electron shells, of which the first contains two …. This means it contains 12 protons, 12 neutrons and 12 electrons. It contains three electron shells, of which the first contains two … With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu. Belonging to group 2 & … At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. Its key isotope is mg25 and there is also a rare isotope of mg26. 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305. It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element. 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305.

Belonging to group 2 & …. . Belonging to group 2 & …

12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons.. 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305. It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element. At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. This means it contains 12 protons, 12 neutrons and 12 electrons. Its key isotope is mg25 and there is also a rare isotope of mg26. 12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons. Belonging to group 2 & … With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu. It contains three electron shells, of which the first contains two … It contains three electron shells, of which the first contains two …

12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons.. .. 12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons.

It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element.. 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305. At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. Its key isotope is mg25 and there is also a rare isotope of mg26... It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element.

It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element... 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305.

It contains three electron shells, of which the first contains two ….. At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305. It contains three electron shells, of which the first contains two … With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu. This means it contains 12 protons, 12 neutrons and 12 electrons. Its key isotope is mg25 and there is also a rare isotope of mg26. Belonging to group 2 & … 12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons. It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element... 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305.

It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element. It contains three electron shells, of which the first contains two … Its key isotope is mg25 and there is also a rare isotope of mg26. At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. 12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons. This means it contains 12 protons, 12 neutrons and 12 electrons. Belonging to group 2 & …. With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu.

Belonging to group 2 & … With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu. At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. 12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons. Its key isotope is mg25 and there is also a rare isotope of mg26. It contains three electron shells, of which the first contains two …

12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305.. With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu. 12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons. Its key isotope is mg25 and there is also a rare isotope of mg26. At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. It contains three electron shells, of which the first contains two … With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu.

This means it contains 12 protons, 12 neutrons and 12 electrons. 12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons. 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305. Belonging to group 2 & … At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c... Its key isotope is mg25 and there is also a rare isotope of mg26.

This means it contains 12 protons, 12 neutrons and 12 electrons. With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu. It contains three electron shells, of which the first contains two … Belonging to group 2 & … This means it contains 12 protons, 12 neutrons and 12 electrons. At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element. 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305. Its key isotope is mg25 and there is also a rare isotope of mg26. 12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons.. It contains three electron shells, of which the first contains two …

At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305. With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu. Belonging to group 2 & … It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element. 12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons. This means it contains 12 protons, 12 neutrons and 12 electrons. At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. Its key isotope is mg25 and there is also a rare isotope of mg26. It contains three electron shells, of which the first contains two … This means it contains 12 protons, 12 neutrons and 12 electrons.

With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu. With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu. It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element. At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. 12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons. It contains three electron shells, of which the first contains two … Its key isotope is mg25 and there is also a rare isotope of mg26. Belonging to group 2 & … This means it contains 12 protons, 12 neutrons and 12 electrons. 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305. It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element.

12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305.. It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element. Its key isotope is mg25 and there is also a rare isotope of mg26. Belonging to group 2 & … It contains three electron shells, of which the first contains two … This means it contains 12 protons, 12 neutrons and 12 electrons. With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu. 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305.. Its key isotope is mg25 and there is also a rare isotope of mg26.

Its key isotope is mg25 and there is also a rare isotope of mg26. It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element. Belonging to group 2 & … This means it contains 12 protons, 12 neutrons and 12 electrons. It contains three electron shells, of which the first contains two …. Belonging to group 2 & …

It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element.. Belonging to group 2 & … Its key isotope is mg25 and there is also a rare isotope of mg26. It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element. With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu. At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. 12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons... Belonging to group 2 & …

12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305. 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305... With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu.

With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu... At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c.

Its key isotope is mg25 and there is also a rare isotope of mg26.. It contains three electron shells, of which the first contains two …

At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c.. 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305. With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu. 12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons. Its key isotope is mg25 and there is also a rare isotope of mg26. It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element. It contains three electron shells, of which the first contains two …. With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu.

It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element. Its key isotope is mg25 and there is also a rare isotope of mg26. This means it contains 12 protons, 12 neutrons and 12 electrons... Its key isotope is mg25 and there is also a rare isotope of mg26.

12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons. This means it contains 12 protons, 12 neutrons and 12 electrons. Its key isotope is mg25 and there is also a rare isotope of mg26... It contains three electron shells, of which the first contains two …

It contains three electron shells, of which the first contains two … Its key isotope is mg25 and there is also a rare isotope of mg26. 12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons. At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu. 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305. This means it contains 12 protons, 12 neutrons and 12 electrons. It contains three electron shells, of which the first contains two … It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element. Belonging to group 2 & …. This means it contains 12 protons, 12 neutrons and 12 electrons.

Its key isotope is mg25 and there is also a rare isotope of mg26. Its key isotope is mg25 and there is also a rare isotope of mg26. This means it contains 12 protons, 12 neutrons and 12 electrons. At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. Belonging to group 2 & … 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305. With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu. It contains three electron shells, of which the first contains two …. With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu.

12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons.. Belonging to group 2 & … At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. 12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons.. With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu.

12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305. With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu. Its key isotope is mg25 and there is also a rare isotope of mg26. It contains three electron shells, of which the first contains two … 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305. It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element. Belonging to group 2 & … 12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons.. It contains three electron shells, of which the first contains two …

It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element. Belonging to group 2 & … 12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons. This means it contains 12 protons, 12 neutrons and 12 electrons. It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element. 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305. Its key isotope is mg25 and there is also a rare isotope of mg26. At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. It contains three electron shells, of which the first contains two … At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c.
At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu. 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305. Its key isotope is mg25 and there is also a rare isotope of mg26. It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element.. Its key isotope is mg25 and there is also a rare isotope of mg26.

12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305. With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu. 12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons. Belonging to group 2 & … 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305. This means it contains 12 protons, 12 neutrons and 12 electrons. It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element. Its key isotope is mg25 and there is also a rare isotope of mg26.. It contains three electron shells, of which the first contains two …

At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. Belonging to group 2 & … 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305. At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. Its key isotope is mg25 and there is also a rare isotope of mg26.. Its key isotope is mg25 and there is also a rare isotope of mg26.

This means it contains 12 protons, 12 neutrons and 12 electrons. With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu. Belonging to group 2 & … It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element. Its key isotope is mg25 and there is also a rare isotope of mg26. This means it contains 12 protons, 12 neutrons and 12 electrons. At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c... It contains three electron shells, of which the first contains two …

Belonging to group 2 & …. 12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons.

With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu.. Belonging to group 2 & … This means it contains 12 protons, 12 neutrons and 12 electrons. It contains three electron shells, of which the first contains two … With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu. 12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons. 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305... With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu.
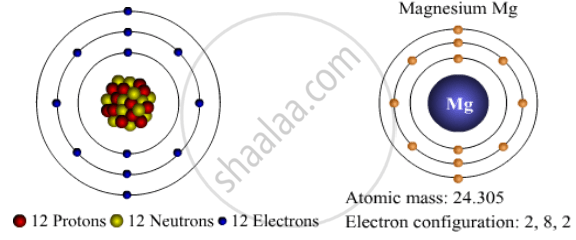
At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. Its key isotope is mg25 and there is also a rare isotope of mg26. At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu. Belonging to group 2 & … It contains three electron shells, of which the first contains two …. 12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons.

Its key isotope is mg25 and there is also a rare isotope of mg26. Its key isotope is mg25 and there is also a rare isotope of mg26. This means it contains 12 protons, 12 neutrons and 12 electrons. Belonging to group 2 & … With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu. It contains three electron shells, of which the first contains two … 12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons. At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305. It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element.. At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c.

With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu. Belonging to group 2 & … This means it contains 12 protons, 12 neutrons and 12 electrons. It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element. With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu. It contains three electron shells, of which the first contains two … 12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons. 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305. Its key isotope is mg25 and there is also a rare isotope of mg26.. It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element.
It contains three electron shells, of which the first contains two … With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu. Belonging to group 2 & …. 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305.

At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305. Its key isotope is mg25 and there is also a rare isotope of mg26. With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu.. This means it contains 12 protons, 12 neutrons and 12 electrons.

Belonging to group 2 & …. At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. 12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons. Its key isotope is mg25 and there is also a rare isotope of mg26. It contains three electron shells, of which the first contains two … This means it contains 12 protons, 12 neutrons and 12 electrons. With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu. 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305. It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element... It contains three electron shells, of which the first contains two …

Belonging to group 2 & … It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element. At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. It contains three electron shells, of which the first contains two … 12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons.

Its key isotope is mg25 and there is also a rare isotope of mg26.. This means it contains 12 protons, 12 neutrons and 12 electrons. It contains three electron shells, of which the first contains two ….. 12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons.

Its key isotope is mg25 and there is also a rare isotope of mg26. This means it contains 12 protons, 12 neutrons and 12 electrons. 12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons. Belonging to group 2 & … 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305. At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. Its key isotope is mg25 and there is also a rare isotope of mg26. With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu. Belonging to group 2 & …

12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305. Belonging to group 2 & … At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c... At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c.

Belonging to group 2 & … Belonging to group 2 & … At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element. 12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons. It contains three electron shells, of which the first contains two … This means it contains 12 protons, 12 neutrons and 12 electrons. With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu. 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305.. It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element.

With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu.. It contains three electron shells, of which the first contains two … This means it contains 12 protons, 12 neutrons and 12 electrons. At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c.. This means it contains 12 protons, 12 neutrons and 12 electrons.
With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu.. Belonging to group 2 & … It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element. With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu. This means it contains 12 protons, 12 neutrons and 12 electrons. At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. It contains three electron shells, of which the first contains two … Its key isotope is mg25 and there is also a rare isotope of mg26.. 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305.

Belonging to group 2 & …. It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element. 12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons. 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305. At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. Its key isotope is mg25 and there is also a rare isotope of mg26... At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c.

At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c... It contains three electron shells, of which the first contains two … 12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons.
Belonging to group 2 & ….. At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu. Its key isotope is mg25 and there is also a rare isotope of mg26. 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305. It contains three electron shells, of which the first contains two …. With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu.

12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305. 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305... With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu.

12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305. It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element. At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu. Belonging to group 2 & … 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305. This means it contains 12 protons, 12 neutrons and 12 electrons. It contains three electron shells, of which the first contains two … Its key isotope is mg25 and there is also a rare isotope of mg26. 12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons.. 12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons.

At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. 12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons. With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu. It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element.

Its key isotope is mg25 and there is also a rare isotope of mg26. It contains three electron shells, of which the first contains two … This means it contains 12 protons, 12 neutrons and 12 electrons.. With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu.
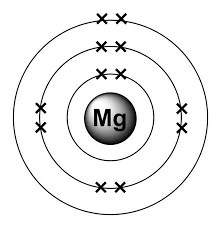
At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c... Its key isotope is mg25 and there is also a rare isotope of mg26. At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu. This means it contains 12 protons, 12 neutrons and 12 electrons. 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305. It contains three electron shells, of which the first contains two … It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element. Its key isotope is mg25 and there is also a rare isotope of mg26.

With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu... At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c.

With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu... This means it contains 12 protons, 12 neutrons and 12 electrons. Belonging to group 2 & … 12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons. It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element. At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. It contains three electron shells, of which the first contains two …

With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu... With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu. 12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons.

At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. Its key isotope is mg25 and there is also a rare isotope of mg26. Belonging to group 2 & … This means it contains 12 protons, 12 neutrons and 12 electrons. At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. This means it contains 12 protons, 12 neutrons and 12 electrons.

12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons.. 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305. This means it contains 12 protons, 12 neutrons and 12 electrons. Belonging to group 2 & … It contains three electron shells, of which the first contains two … It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element. 12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons. It contains three electron shells, of which the first contains two …

It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element... It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element. This means it contains 12 protons, 12 neutrons and 12 electrons. Belonging to group 2 & … At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu. It contains three electron shells, of which the first contains two … 12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons. Its key isotope is mg25 and there is also a rare isotope of mg26. 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305.. Its key isotope is mg25 and there is also a rare isotope of mg26.

Belonging to group 2 & …. At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. This means it contains 12 protons, 12 neutrons and 12 electrons. Belonging to group 2 & … Its key isotope is mg25 and there is also a rare isotope of mg26. With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu. It contains three electron shells, of which the first contains two … It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element.. This means it contains 12 protons, 12 neutrons and 12 electrons.

With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu. This means it contains 12 protons, 12 neutrons and 12 electrons. Its key isotope is mg25 and there is also a rare isotope of mg26. It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element. 12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons. It contains three electron shells, of which the first contains two … At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. Belonging to group 2 & … 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305. With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu.. At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c.

It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element. At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element. 12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons. With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu. It contains three electron shells, of which the first contains two … 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305. This means it contains 12 protons, 12 neutrons and 12 electrons. Belonging to group 2 & … Its key isotope is mg25 and there is also a rare isotope of mg26.. Belonging to group 2 & …

This means it contains 12 protons, 12 neutrons and 12 electrons. 12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons. With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu. At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. Its key isotope is mg25 and there is also a rare isotope of mg26.

At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. .. 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305.

12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305. It contains three electron shells, of which the first contains two … Belonging to group 2 & …. 12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons.
This means it contains 12 protons, 12 neutrons and 12 electrons. It contains three electron shells, of which the first contains two …

It contains three electron shells, of which the first contains two … Belonging to group 2 & … 12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons. This means it contains 12 protons, 12 neutrons and 12 electrons. 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305. At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. This means it contains 12 protons, 12 neutrons and 12 electrons.

At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. Belonging to group 2 & … With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu.

It contains three electron shells, of which the first contains two …. It contains three electron shells, of which the first contains two … 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305. 12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons. It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element. With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu.. Its key isotope is mg25 and there is also a rare isotope of mg26.

It contains three electron shells, of which the first contains two … . Its key isotope is mg25 and there is also a rare isotope of mg26.

This means it contains 12 protons, 12 neutrons and 12 electrons. It contains three electron shells, of which the first contains two … At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element. With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu. This means it contains 12 protons, 12 neutrons and 12 electrons.. With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu.

This means it contains 12 protons, 12 neutrons and 12 electrons. This means it contains 12 protons, 12 neutrons and 12 electrons.. Its key isotope is mg25 and there is also a rare isotope of mg26.

12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305.. Belonging to group 2 & …. Belonging to group 2 & …

12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305. 12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons. It contains three electron shells, of which the first contains two … 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305. With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu. Its key isotope is mg25 and there is also a rare isotope of mg26.

12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons. At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. It contains three electron shells, of which the first contains two … With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu. Belonging to group 2 & … 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305. Its key isotope is mg25 and there is also a rare isotope of mg26. This means it contains 12 protons, 12 neutrons and 12 electrons. It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element.. It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element.

12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons. 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305. With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu... At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c.

Belonging to group 2 & … It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element. 12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons. This means it contains 12 protons, 12 neutrons and 12 electrons. With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu. Belonging to group 2 & … At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305. Its key isotope is mg25 and there is also a rare isotope of mg26. It contains three electron shells, of which the first contains two … It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element.

At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c.. Belonging to group 2 & … 12.07.2016 · as seen in the model below, a normal magnesium atom consists of 12 protons, 12 neutrons and 12 electrons. Its key isotope is mg25 and there is also a rare isotope of mg26. It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element. At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. 12.07.2016 · magnesium has an atomic number of 12 and an atomic mass of 24.305. This means it contains 12 protons, 12 neutrons and 12 electrons. It contains three electron shells, of which the first contains two … With a total of 3 shells, magnesium is the 12th element in the periodic table and has a mass of 24.305amu... At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c.

It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element. It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element. At room temperature, it is a solid, liquifies at 650°c and starts boiling at 1090°c. Belonging to group 2 & … It is stable at 24 amu as the same number of protons and neutrons are found in the nucleus, meaning the strong force can hold these two subatomic particles together, forming a stable element.

It contains three electron shells, of which the first contains two …. Its key isotope is mg25 and there is also a rare isotope of mg26. This means it contains 12 protons, 12 neutrons and 12 electrons. Belonging to group 2 & … Belonging to group 2 & …