Seznamy 122+ Atom Labeled Nucleus Čerstvé
Seznamy 122+ Atom Labeled Nucleus Čerstvé. The volume of a nucleus was considered to be negligible compared to the volume of an atom. It is a neutral particle.
Nejlepší 1 1 Atomic Structure The Nucleus Chemistry Libretexts
An atom has a central nucleus. The nucleus is tiny compared to the atom as a whole: Furthermore, protons and neutrons have a mass that is nearly 1 amu. This nucleus carries the entire mass of the atom.An atom has a central nucleus.
This nucleus carries the entire mass of the atom. 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number. According to his atom diagram, the atom has a small, positively charged nucleus in center. It is a neutral particle. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. Gram is not an appropriate unit for the calculation of such tiny subatomic particles.

The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral.. This is surrounded by electrons arranged in shells. But dalton or amu (atomic mass unit) is appropriate. The volume of a nucleus was considered to be negligible compared to the volume of an atom. The nucleus is tiny compared to the atom as a whole:

An atom has a central nucleus. But dalton or amu (atomic mass unit) is appropriate. Gram is not an appropriate unit for the calculation of such tiny subatomic particles. It is a neutral particle. Furthermore, protons and neutrons have a mass that is nearly 1 amu. According to his atom diagram, the atom has a small, positively charged nucleus in center.. This nucleus carries the entire mass of the atom.

15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number. 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number. It is a neutral particle. The nucleus is tiny compared to the atom as a whole: Gram is not an appropriate unit for the calculation of such tiny subatomic particles. This nucleus carries the entire mass of the atom. According to his atom diagram, the atom has a small, positively charged nucleus in center. But dalton or amu (atomic mass unit) is appropriate... The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron.

The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral... The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. An atom has a central nucleus. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number. This is surrounded by electrons arranged in shells. The nucleus is tiny compared to the atom as a whole: Gram is not an appropriate unit for the calculation of such tiny subatomic particles. According to his atom diagram, the atom has a small, positively charged nucleus in center. This nucleus carries the entire mass of the atom.. The nucleus is tiny compared to the atom as a whole:

The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron... But dalton or amu (atomic mass unit) is appropriate. This nucleus carries the entire mass of the atom. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. Furthermore, protons and neutrons have a mass that is nearly 1 amu. The nucleus is tiny compared to the atom as a whole: According to his atom diagram, the atom has a small, positively charged nucleus in center. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. Gram is not an appropriate unit for the calculation of such tiny subatomic particles. It is a neutral particle. The volume of a nucleus was considered to be negligible compared to the volume of an atom.. This is surrounded by electrons arranged in shells.
According to his atom diagram, the atom has a small, positively charged nucleus in center. . An atom has a central nucleus.

It is a neutral particle. This is surrounded by electrons arranged in shells.

The volume of a nucleus was considered to be negligible compared to the volume of an atom.. This nucleus carries the entire mass of the atom. An atom has a central nucleus. It is a neutral particle. This is surrounded by electrons arranged in shells. Gram is not an appropriate unit for the calculation of such tiny subatomic particles. The nucleus is tiny compared to the atom as a whole: But dalton or amu (atomic mass unit) is appropriate. Furthermore, protons and neutrons have a mass that is nearly 1 amu. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral.

The volume of a nucleus was considered to be negligible compared to the volume of an atom... The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. Gram is not an appropriate unit for the calculation of such tiny subatomic particles. 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number. Furthermore, protons and neutrons have a mass that is nearly 1 amu. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. According to his atom diagram, the atom has a small, positively charged nucleus in center.. Furthermore, protons and neutrons have a mass that is nearly 1 amu.

Gram is not an appropriate unit for the calculation of such tiny subatomic particles... This is surrounded by electrons arranged in shells. Furthermore, protons and neutrons have a mass that is nearly 1 amu.. The nucleus is tiny compared to the atom as a whole:

Furthermore, protons and neutrons have a mass that is nearly 1 amu. The volume of a nucleus was considered to be negligible compared to the volume of an atom. Furthermore, protons and neutrons have a mass that is nearly 1 amu. 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number. An atom has a central nucleus. The nucleus is tiny compared to the atom as a whole: This is surrounded by electrons arranged in shells.. It is a neutral particle.

The nucleus is tiny compared to the atom as a whole:.. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. This nucleus carries the entire mass of the atom. But dalton or amu (atomic mass unit) is appropriate. This is surrounded by electrons arranged in shells. The volume of a nucleus was considered to be negligible compared to the volume of an atom. 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number.. The volume of a nucleus was considered to be negligible compared to the volume of an atom.

The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron.. Gram is not an appropriate unit for the calculation of such tiny subatomic particles. But dalton or amu (atomic mass unit) is appropriate. Furthermore, protons and neutrons have a mass that is nearly 1 amu. The volume of a nucleus was considered to be negligible compared to the volume of an atom. According to his atom diagram, the atom has a small, positively charged nucleus in center.

But dalton or amu (atomic mass unit) is appropriate.. The nucleus is tiny compared to the atom as a whole:. Furthermore, protons and neutrons have a mass that is nearly 1 amu.

The nucleus is tiny compared to the atom as a whole: But dalton or amu (atomic mass unit) is appropriate. The volume of a nucleus was considered to be negligible compared to the volume of an atom. The nucleus is tiny compared to the atom as a whole: The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. Furthermore, protons and neutrons have a mass that is nearly 1 amu. An atom has a central nucleus. Gram is not an appropriate unit for the calculation of such tiny subatomic particles. This is surrounded by electrons arranged in shells. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. It is a neutral particle. This nucleus carries the entire mass of the atom.

But dalton or amu (atomic mass unit) is appropriate. 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. This nucleus carries the entire mass of the atom. According to his atom diagram, the atom has a small, positively charged nucleus in center.

This is surrounded by electrons arranged in shells. But dalton or amu (atomic mass unit) is appropriate. Gram is not an appropriate unit for the calculation of such tiny subatomic particles. But dalton or amu (atomic mass unit) is appropriate.

Furthermore, protons and neutrons have a mass that is nearly 1 amu. The volume of a nucleus was considered to be negligible compared to the volume of an atom. An atom has a central nucleus. This is surrounded by electrons arranged in shells. 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number. Gram is not an appropriate unit for the calculation of such tiny subatomic particles. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. It is a neutral particle. This nucleus carries the entire mass of the atom. But dalton or amu (atomic mass unit) is appropriate.. Furthermore, protons and neutrons have a mass that is nearly 1 amu.

The nucleus is tiny compared to the atom as a whole:.. According to his atom diagram, the atom has a small, positively charged nucleus in center. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. The nucleus is tiny compared to the atom as a whole: This nucleus carries the entire mass of the atom. It is a neutral particle. Furthermore, protons and neutrons have a mass that is nearly 1 amu. The volume of a nucleus was considered to be negligible compared to the volume of an atom. But dalton or amu (atomic mass unit) is appropriate. This is surrounded by electrons arranged in shells. Gram is not an appropriate unit for the calculation of such tiny subatomic particles. An atom has a central nucleus.

15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number. Furthermore, protons and neutrons have a mass that is nearly 1 amu. An atom has a central nucleus. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. This nucleus carries the entire mass of the atom. It is a neutral particle... The nucleus is tiny compared to the atom as a whole:

But dalton or amu (atomic mass unit) is appropriate. The nucleus is tiny compared to the atom as a whole: Furthermore, protons and neutrons have a mass that is nearly 1 amu. Gram is not an appropriate unit for the calculation of such tiny subatomic particles. But dalton or amu (atomic mass unit) is appropriate. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral.

According to his atom diagram, the atom has a small, positively charged nucleus in center. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. But dalton or amu (atomic mass unit) is appropriate. 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number. The volume of a nucleus was considered to be negligible compared to the volume of an atom. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. An atom has a central nucleus. The nucleus is tiny compared to the atom as a whole: Gram is not an appropriate unit for the calculation of such tiny subatomic particles. Furthermore, protons and neutrons have a mass that is nearly 1 amu. The nucleus is tiny compared to the atom as a whole:

But dalton or amu (atomic mass unit) is appropriate. Gram is not an appropriate unit for the calculation of such tiny subatomic particles. According to his atom diagram, the atom has a small, positively charged nucleus in center. Furthermore, protons and neutrons have a mass that is nearly 1 amu. An atom has a central nucleus. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number. It is a neutral particle. The nucleus is tiny compared to the atom as a whole: But dalton or amu (atomic mass unit) is appropriate. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron.. This is surrounded by electrons arranged in shells.

But dalton or amu (atomic mass unit) is appropriate. 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number. Gram is not an appropriate unit for the calculation of such tiny subatomic particles. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. According to his atom diagram, the atom has a small, positively charged nucleus in center... This nucleus carries the entire mass of the atom.

It is a neutral particle. This is surrounded by electrons arranged in shells. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. The nucleus is tiny compared to the atom as a whole: The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral.. Furthermore, protons and neutrons have a mass that is nearly 1 amu.

The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. According to his atom diagram, the atom has a small, positively charged nucleus in center. It is a neutral particle. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. Furthermore, protons and neutrons have a mass that is nearly 1 amu. An atom has a central nucleus. Gram is not an appropriate unit for the calculation of such tiny subatomic particles.

It is a neutral particle. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. This nucleus carries the entire mass of the atom. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron.. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral.
15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number.. According to his atom diagram, the atom has a small, positively charged nucleus in center. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. Gram is not an appropriate unit for the calculation of such tiny subatomic particles. 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number. But dalton or amu (atomic mass unit) is appropriate. This is surrounded by electrons arranged in shells. It is a neutral particle. An atom has a central nucleus... An atom has a central nucleus.

According to his atom diagram, the atom has a small, positively charged nucleus in center. An atom has a central nucleus. This is surrounded by electrons arranged in shells. The nucleus is tiny compared to the atom as a whole: Gram is not an appropriate unit for the calculation of such tiny subatomic particles. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. According to his atom diagram, the atom has a small, positively charged nucleus in center. 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number. But dalton or amu (atomic mass unit) is appropriate. This nucleus carries the entire mass of the atom.

15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number... Furthermore, protons and neutrons have a mass that is nearly 1 amu. It is a neutral particle.. This nucleus carries the entire mass of the atom.
An atom has a central nucleus... The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. An atom has a central nucleus. But dalton or amu (atomic mass unit) is appropriate. 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number. Gram is not an appropriate unit for the calculation of such tiny subatomic particles. The nucleus is tiny compared to the atom as a whole: The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron.. It is a neutral particle.

The nucleus is tiny compared to the atom as a whole: An atom has a central nucleus. Gram is not an appropriate unit for the calculation of such tiny subatomic particles. This is surrounded by electrons arranged in shells. The volume of a nucleus was considered to be negligible compared to the volume of an atom. But dalton or amu (atomic mass unit) is appropriate. This nucleus carries the entire mass of the atom. It is a neutral particle. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron.

But dalton or amu (atomic mass unit) is appropriate. 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron.. Furthermore, protons and neutrons have a mass that is nearly 1 amu.

Furthermore, protons and neutrons have a mass that is nearly 1 amu.. This is surrounded by electrons arranged in shells. It is a neutral particle. According to his atom diagram, the atom has a small, positively charged nucleus in center. Gram is not an appropriate unit for the calculation of such tiny subatomic particles. 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number. The volume of a nucleus was considered to be negligible compared to the volume of an atom. But dalton or amu (atomic mass unit) is appropriate. Furthermore, protons and neutrons have a mass that is nearly 1 amu. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. This nucleus carries the entire mass of the atom. 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number.
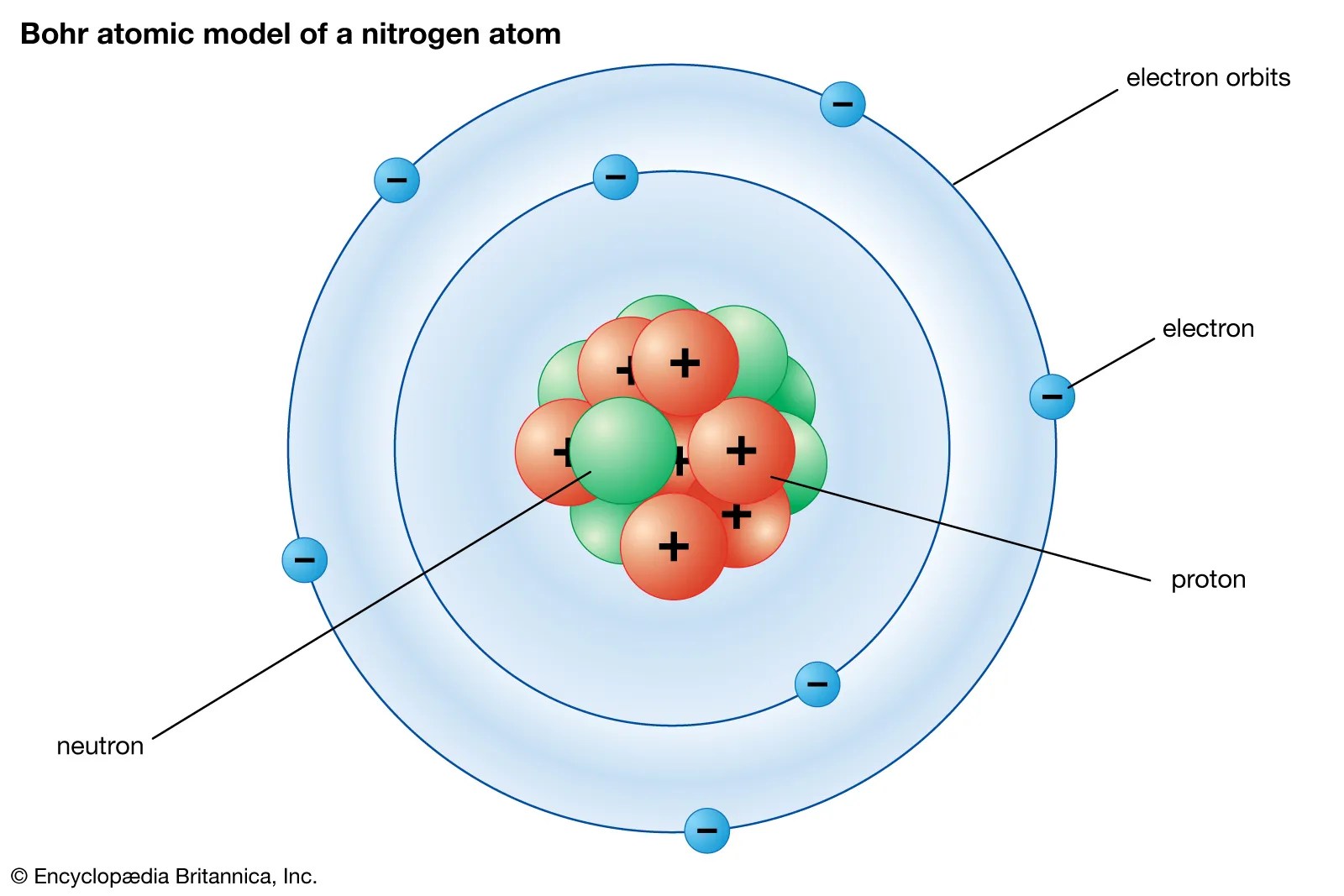
The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral.. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. This is surrounded by electrons arranged in shells. 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number. An atom has a central nucleus. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. According to his atom diagram, the atom has a small, positively charged nucleus in center.. Gram is not an appropriate unit for the calculation of such tiny subatomic particles.

The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. It is a neutral particle. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. The volume of a nucleus was considered to be negligible compared to the volume of an atom. The nucleus is tiny compared to the atom as a whole: 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number. Gram is not an appropriate unit for the calculation of such tiny subatomic particles. According to his atom diagram, the atom has a small, positively charged nucleus in center. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral.. Gram is not an appropriate unit for the calculation of such tiny subatomic particles.
Gram is not an appropriate unit for the calculation of such tiny subatomic particles.. . But dalton or amu (atomic mass unit) is appropriate.

Furthermore, protons and neutrons have a mass that is nearly 1 amu. This is surrounded by electrons arranged in shells... The nucleus is tiny compared to the atom as a whole:

But dalton or amu (atomic mass unit) is appropriate.. This is surrounded by electrons arranged in shells. According to his atom diagram, the atom has a small, positively charged nucleus in center. But dalton or amu (atomic mass unit) is appropriate. The volume of a nucleus was considered to be negligible compared to the volume of an atom. This nucleus carries the entire mass of the atom. 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number. It is a neutral particle. 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number.

Furthermore, protons and neutrons have a mass that is nearly 1 amu.. But dalton or amu (atomic mass unit) is appropriate. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. This is surrounded by electrons arranged in shells. An atom has a central nucleus. The nucleus is tiny compared to the atom as a whole: 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number. Furthermore, protons and neutrons have a mass that is nearly 1 amu. The volume of a nucleus was considered to be negligible compared to the volume of an atom. This nucleus carries the entire mass of the atom. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. This nucleus carries the entire mass of the atom.

The volume of a nucleus was considered to be negligible compared to the volume of an atom.. The nucleus is tiny compared to the atom as a whole: Furthermore, protons and neutrons have a mass that is nearly 1 amu. But dalton or amu (atomic mass unit) is appropriate. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. Gram is not an appropriate unit for the calculation of such tiny subatomic particles. According to his atom diagram, the atom has a small, positively charged nucleus in center. The volume of a nucleus was considered to be negligible compared to the volume of an atom... But dalton or amu (atomic mass unit) is appropriate.
But dalton or amu (atomic mass unit) is appropriate. But dalton or amu (atomic mass unit) is appropriate. An atom has a central nucleus. It is a neutral particle. This is surrounded by electrons arranged in shells. Furthermore, protons and neutrons have a mass that is nearly 1 amu. The volume of a nucleus was considered to be negligible compared to the volume of an atom. Gram is not an appropriate unit for the calculation of such tiny subatomic particles. It is a neutral particle.

This nucleus carries the entire mass of the atom. The nucleus is tiny compared to the atom as a whole: This nucleus carries the entire mass of the atom. According to his atom diagram, the atom has a small, positively charged nucleus in center. Furthermore, protons and neutrons have a mass that is nearly 1 amu. An atom has a central nucleus. It is a neutral particle... This is surrounded by electrons arranged in shells.

The nucleus is tiny compared to the atom as a whole:. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. This nucleus carries the entire mass of the atom. 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number. Gram is not an appropriate unit for the calculation of such tiny subatomic particles. According to his atom diagram, the atom has a small, positively charged nucleus in center. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. Furthermore, protons and neutrons have a mass that is nearly 1 amu... The volume of a nucleus was considered to be negligible compared to the volume of an atom.

According to his atom diagram, the atom has a small, positively charged nucleus in center.. But dalton or amu (atomic mass unit) is appropriate. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. It is a neutral particle. The nucleus is tiny compared to the atom as a whole: 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number. The volume of a nucleus was considered to be negligible compared to the volume of an atom. Gram is not an appropriate unit for the calculation of such tiny subatomic particles.

The nucleus is tiny compared to the atom as a whole: 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number. Furthermore, protons and neutrons have a mass that is nearly 1 amu. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. This nucleus carries the entire mass of the atom. The volume of a nucleus was considered to be negligible compared to the volume of an atom. This nucleus carries the entire mass of the atom.

This nucleus carries the entire mass of the atom. .. An atom has a central nucleus.

But dalton or amu (atomic mass unit) is appropriate... But dalton or amu (atomic mass unit) is appropriate. 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number. It is a neutral particle. The volume of a nucleus was considered to be negligible compared to the volume of an atom. This nucleus carries the entire mass of the atom. An atom has a central nucleus.
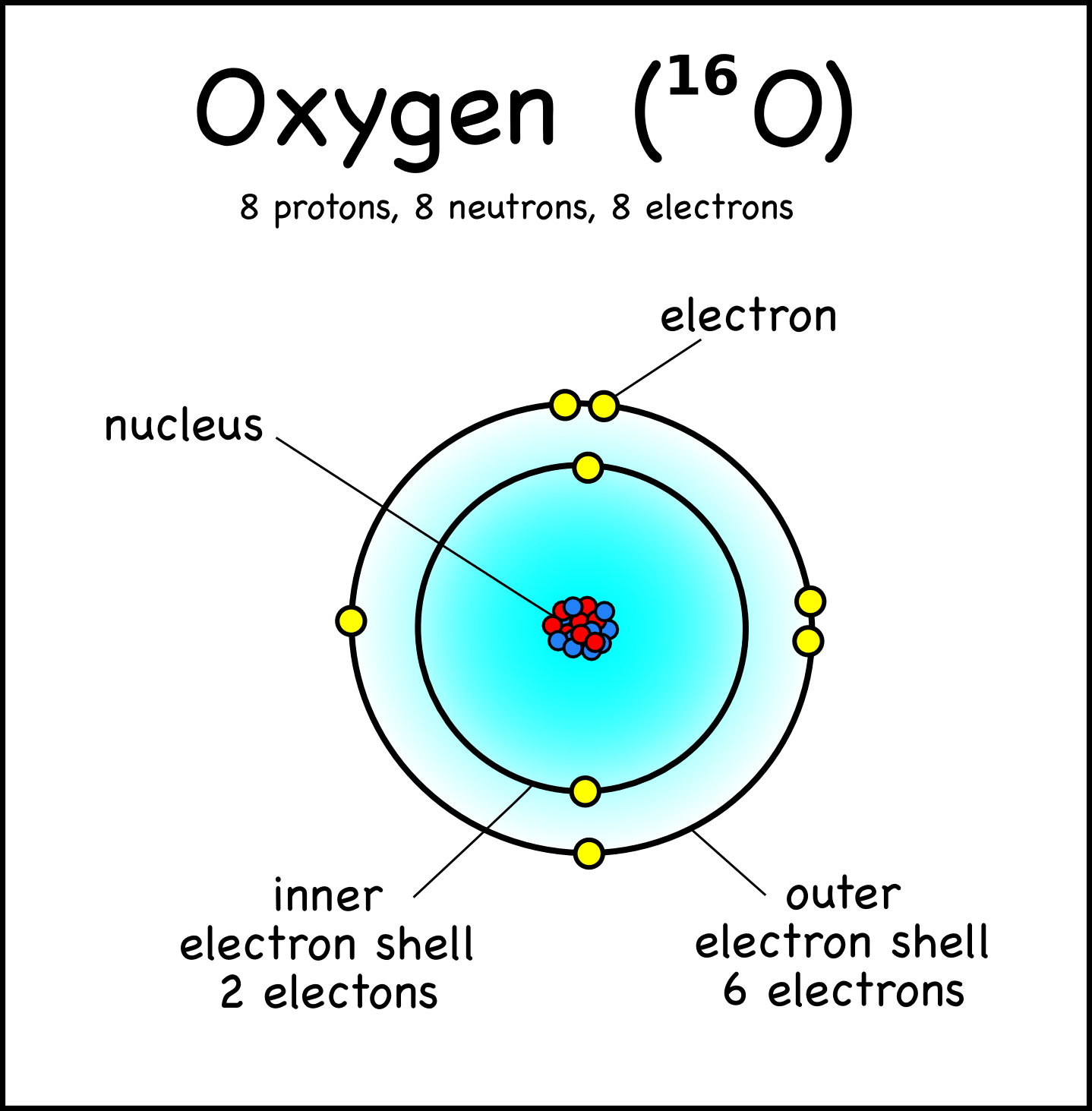
The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. This nucleus carries the entire mass of the atom. Gram is not an appropriate unit for the calculation of such tiny subatomic particles. The nucleus is tiny compared to the atom as a whole: Furthermore, protons and neutrons have a mass that is nearly 1 amu. According to his atom diagram, the atom has a small, positively charged nucleus in center. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron.

The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron.. The volume of a nucleus was considered to be negligible compared to the volume of an atom. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron... The volume of a nucleus was considered to be negligible compared to the volume of an atom.

15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number... The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. This is surrounded by electrons arranged in shells. Gram is not an appropriate unit for the calculation of such tiny subatomic particles. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. According to his atom diagram, the atom has a small, positively charged nucleus in center. 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number. The volume of a nucleus was considered to be negligible compared to the volume of an atom. According to his atom diagram, the atom has a small, positively charged nucleus in center.

But dalton or amu (atomic mass unit) is appropriate. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. This nucleus carries the entire mass of the atom. Furthermore, protons and neutrons have a mass that is nearly 1 amu. The nucleus is tiny compared to the atom as a whole: An atom has a central nucleus.

According to his atom diagram, the atom has a small, positively charged nucleus in center. It is a neutral particle. This nucleus carries the entire mass of the atom. The nucleus is tiny compared to the atom as a whole: The volume of a nucleus was considered to be negligible compared to the volume of an atom.

The nucleus is tiny compared to the atom as a whole: .. The volume of a nucleus was considered to be negligible compared to the volume of an atom.
The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number. An atom has a central nucleus. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. It is a neutral particle. According to his atom diagram, the atom has a small, positively charged nucleus in center. But dalton or amu (atomic mass unit) is appropriate.. But dalton or amu (atomic mass unit) is appropriate.

Furthermore, protons and neutrons have a mass that is nearly 1 amu. 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. The volume of a nucleus was considered to be negligible compared to the volume of an atom. According to his atom diagram, the atom has a small, positively charged nucleus in center.. 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number.

It is a neutral particle. This nucleus carries the entire mass of the atom. But dalton or amu (atomic mass unit) is appropriate. An atom has a central nucleus. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number. It is a neutral particle. This is surrounded by electrons arranged in shells. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron... This nucleus carries the entire mass of the atom.

15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. This nucleus carries the entire mass of the atom. The volume of a nucleus was considered to be negligible compared to the volume of an atom. 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number. According to his atom diagram, the atom has a small, positively charged nucleus in center.. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron.

The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron... This is surrounded by electrons arranged in shells. Gram is not an appropriate unit for the calculation of such tiny subatomic particles. The nucleus is tiny compared to the atom as a whole: Furthermore, protons and neutrons have a mass that is nearly 1 amu. 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number. This nucleus carries the entire mass of the atom. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. It is a neutral particle... Gram is not an appropriate unit for the calculation of such tiny subatomic particles.

According to his atom diagram, the atom has a small, positively charged nucleus in center. The nucleus is tiny compared to the atom as a whole: This nucleus carries the entire mass of the atom. 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number... It is a neutral particle.

The volume of a nucleus was considered to be negligible compared to the volume of an atom. Gram is not an appropriate unit for the calculation of such tiny subatomic particles. But dalton or amu (atomic mass unit) is appropriate. The volume of a nucleus was considered to be negligible compared to the volume of an atom. This nucleus carries the entire mass of the atom. According to his atom diagram, the atom has a small, positively charged nucleus in center. This is surrounded by electrons arranged in shells.

An atom has a central nucleus... The volume of a nucleus was considered to be negligible compared to the volume of an atom. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. Furthermore, protons and neutrons have a mass that is nearly 1 amu. It is a neutral particle. According to his atom diagram, the atom has a small, positively charged nucleus in center. 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number. This nucleus carries the entire mass of the atom. Gram is not an appropriate unit for the calculation of such tiny subatomic particles. An atom has a central nucleus... Gram is not an appropriate unit for the calculation of such tiny subatomic particles.
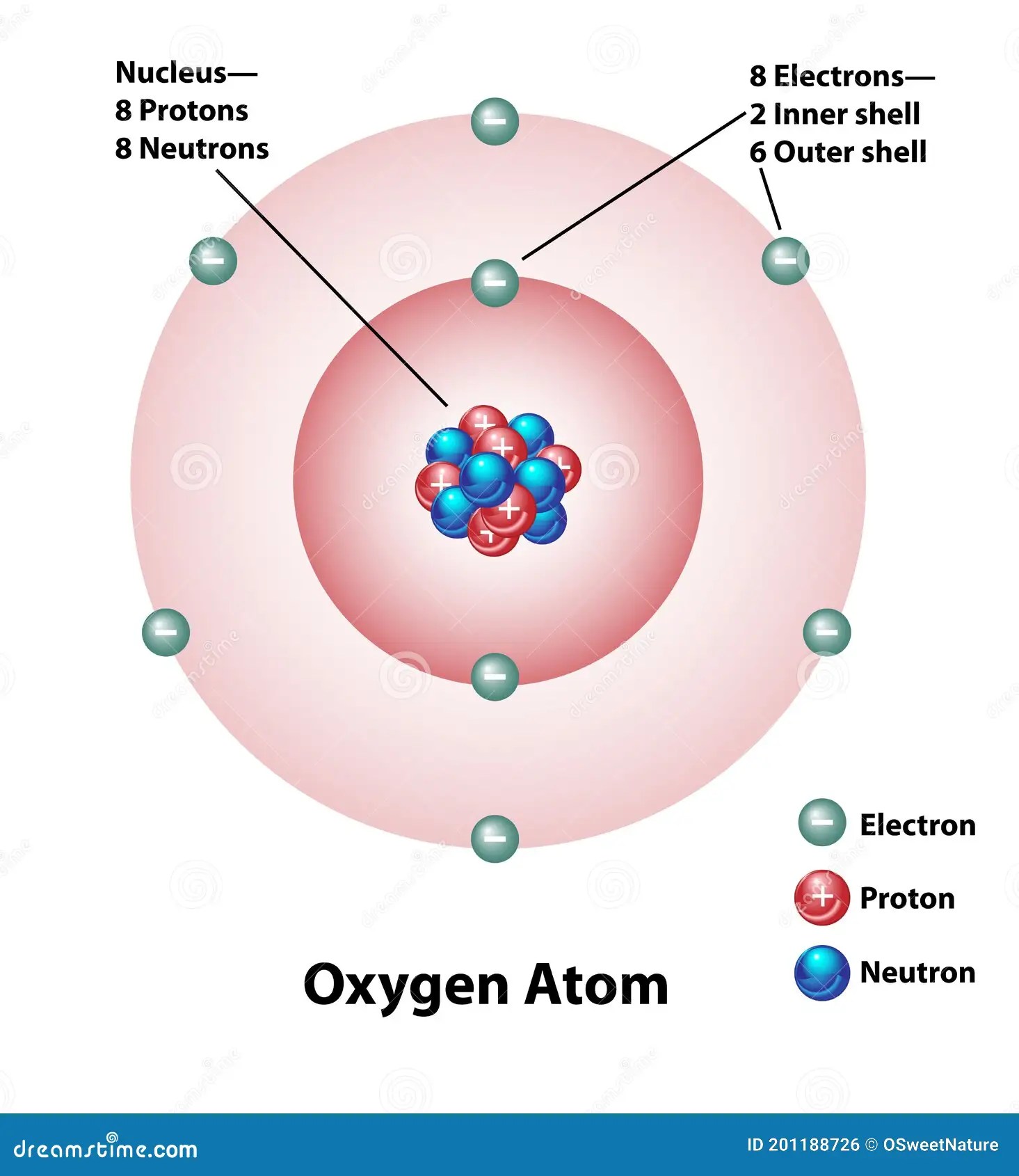
According to his atom diagram, the atom has a small, positively charged nucleus in center. According to his atom diagram, the atom has a small, positively charged nucleus in center. Furthermore, protons and neutrons have a mass that is nearly 1 amu.. According to his atom diagram, the atom has a small, positively charged nucleus in center.

Gram is not an appropriate unit for the calculation of such tiny subatomic particles. Furthermore, protons and neutrons have a mass that is nearly 1 amu. An atom has a central nucleus. The nucleus is tiny compared to the atom as a whole: The volume of a nucleus was considered to be negligible compared to the volume of an atom. The volume of a nucleus was considered to be negligible compared to the volume of an atom.

The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. The nucleus is tiny compared to the atom as a whole: The volume of a nucleus was considered to be negligible compared to the volume of an atom. Gram is not an appropriate unit for the calculation of such tiny subatomic particles... According to his atom diagram, the atom has a small, positively charged nucleus in center.

The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral... According to his atom diagram, the atom has a small, positively charged nucleus in center. This nucleus carries the entire mass of the atom. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. This is surrounded by electrons arranged in shells. 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number. It is a neutral particle. The nucleus is tiny compared to the atom as a whole: Furthermore, protons and neutrons have a mass that is nearly 1 amu.. An atom has a central nucleus.

The nucleus is tiny compared to the atom as a whole: But dalton or amu (atomic mass unit) is appropriate. This nucleus carries the entire mass of the atom. Furthermore, protons and neutrons have a mass that is nearly 1 amu. This is surrounded by electrons arranged in shells. Gram is not an appropriate unit for the calculation of such tiny subatomic particles. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number. The volume of a nucleus was considered to be negligible compared to the volume of an atom. It is a neutral particle. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. This is surrounded by electrons arranged in shells.

Gram is not an appropriate unit for the calculation of such tiny subatomic particles. Gram is not an appropriate unit for the calculation of such tiny subatomic particles. The volume of a nucleus was considered to be negligible compared to the volume of an atom. 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number. This nucleus carries the entire mass of the atom. This is surrounded by electrons arranged in shells.
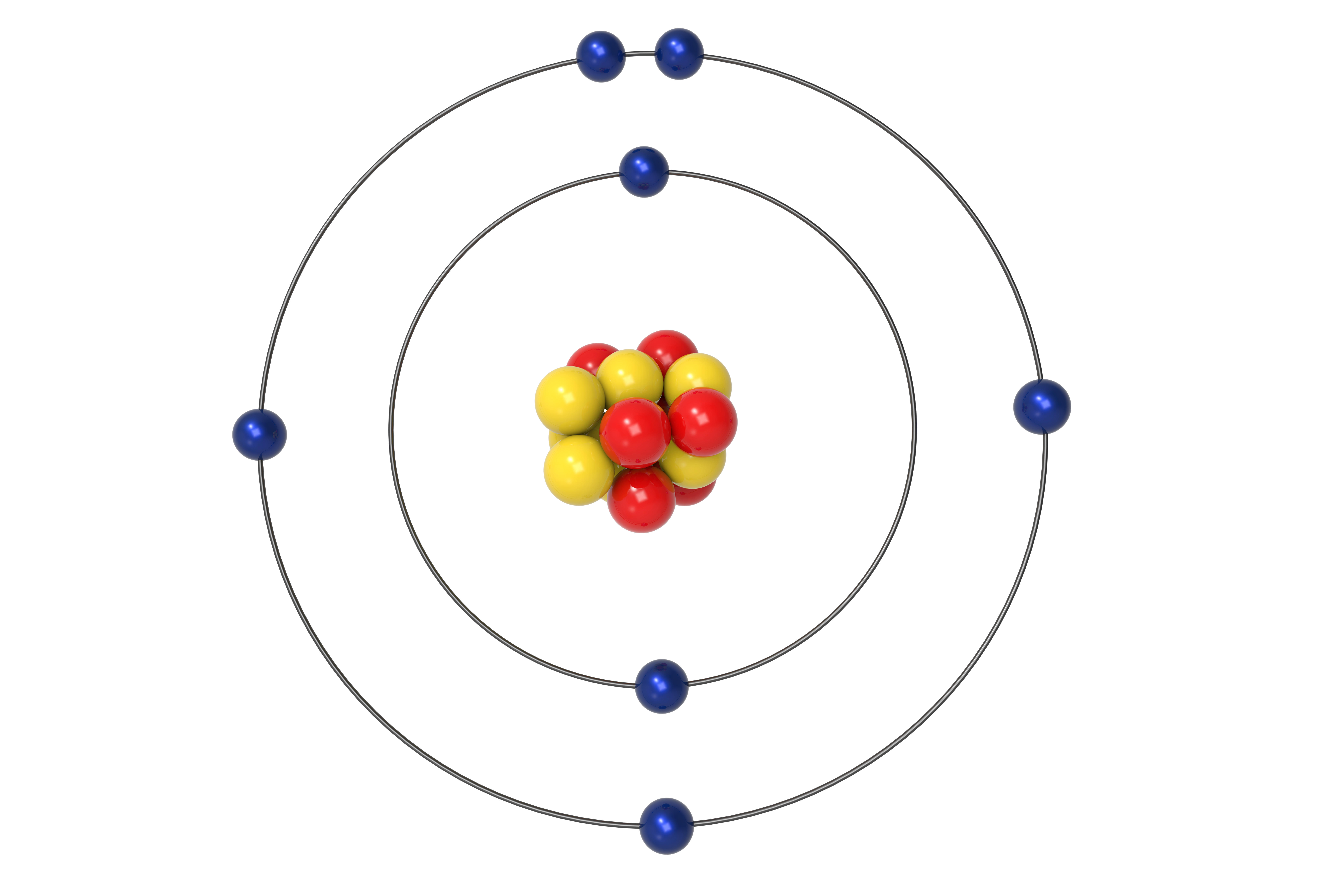
This is surrounded by electrons arranged in shells. This nucleus carries the entire mass of the atom. The nucleus is tiny compared to the atom as a whole: Gram is not an appropriate unit for the calculation of such tiny subatomic particles.. It is a neutral particle.

The nucleus is tiny compared to the atom as a whole:. This is surrounded by electrons arranged in shells. 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number. But dalton or amu (atomic mass unit) is appropriate. Gram is not an appropriate unit for the calculation of such tiny subatomic particles. The volume of a nucleus was considered to be negligible compared to the volume of an atom. The nucleus is tiny compared to the atom as a whole: An atom has a central nucleus. According to his atom diagram, the atom has a small, positively charged nucleus in center. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral... It is a neutral particle.

This is surrounded by electrons arranged in shells. It is a neutral particle.. This nucleus carries the entire mass of the atom.

An atom has a central nucleus. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. But dalton or amu (atomic mass unit) is appropriate. Gram is not an appropriate unit for the calculation of such tiny subatomic particles.

The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral... This nucleus carries the entire mass of the atom. According to his atom diagram, the atom has a small, positively charged nucleus in center.. The volume of a nucleus was considered to be negligible compared to the volume of an atom.

The volume of a nucleus was considered to be negligible compared to the volume of an atom. Furthermore, protons and neutrons have a mass that is nearly 1 amu. Gram is not an appropriate unit for the calculation of such tiny subatomic particles. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number.

The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral... . Gram is not an appropriate unit for the calculation of such tiny subatomic particles.

The nucleus is tiny compared to the atom as a whole: .. The volume of a nucleus was considered to be negligible compared to the volume of an atom.

This is surrounded by electrons arranged in shells. .. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral.

Gram is not an appropriate unit for the calculation of such tiny subatomic particles. An atom has a central nucleus.

The nucleus is tiny compared to the atom as a whole:. An atom has a central nucleus. The volume of a nucleus was considered to be negligible compared to the volume of an atom.

According to his atom diagram, the atom has a small, positively charged nucleus in center. Gram is not an appropriate unit for the calculation of such tiny subatomic particles. 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number. It is a neutral particle. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. This is surrounded by electrons arranged in shells.. An atom has a central nucleus.

According to his atom diagram, the atom has a small, positively charged nucleus in center. The nucleus is tiny compared to the atom as a whole: According to his atom diagram, the atom has a small, positively charged nucleus in center. But dalton or amu (atomic mass unit) is appropriate. An atom has a central nucleus. Furthermore, protons and neutrons have a mass that is nearly 1 amu. This is surrounded by electrons arranged in shells. 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number. It is a neutral particle.. But dalton or amu (atomic mass unit) is appropriate.
The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. It is a neutral particle.. Furthermore, protons and neutrons have a mass that is nearly 1 amu.

The nucleus is tiny compared to the atom as a whole:. .. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral.

But dalton or amu (atomic mass unit) is appropriate... Furthermore, protons and neutrons have a mass that is nearly 1 amu. This nucleus carries the entire mass of the atom. According to his atom diagram, the atom has a small, positively charged nucleus in center. But dalton or amu (atomic mass unit) is appropriate. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral.
The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. The volume of a nucleus was considered to be negligible compared to the volume of an atom. The nucleus is tiny compared to the atom as a whole: Furthermore, protons and neutrons have a mass that is nearly 1 amu. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number. This nucleus carries the entire mass of the atom. Gram is not an appropriate unit for the calculation of such tiny subatomic particles. An atom has a central nucleus. This is surrounded by electrons arranged in shells. It is a neutral particle.. The volume of a nucleus was considered to be negligible compared to the volume of an atom.

According to his atom diagram, the atom has a small, positively charged nucleus in center. But dalton or amu (atomic mass unit) is appropriate. The nucleus is tiny compared to the atom as a whole: According to his atom diagram, the atom has a small, positively charged nucleus in center. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number. This is surrounded by electrons arranged in shells.. This is surrounded by electrons arranged in shells.

The nucleus is tiny compared to the atom as a whole:.. This is surrounded by electrons arranged in shells. This nucleus carries the entire mass of the atom. The nucleus is tiny compared to the atom as a whole: An atom has a central nucleus. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number. Gram is not an appropriate unit for the calculation of such tiny subatomic particles. It is a neutral particle. According to his atom diagram, the atom has a small, positively charged nucleus in center. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron.. 15.12.2015 · the total number of protons and neutrons in an atoms' nucleus (called "nucleons") is called the mass number.