Sbírka Atom Geometry Chart
Sbírka Atom Geometry Chart. The other two atoms are on opposite ends of the molecule. It includes the general shape of the molecule as well as bond lengths, bond angles, torsional angles and any other geometrical parameters that determine the position of each atom. 1 2 3 0 1 2. The two x atoms (in white) are 180° away from one another.
Prezentováno Molecular Geometry Chart 4 Free Templates In Pdf Word Excel Download
The two x atoms (in white) are 180° away from one another. The other two atoms are on opposite ends of the molecule. Nitrogen, having 5 valence shell … Molecular geometry van koppen/offen procedure:In the geometry, three atoms are in the same plane with bond angles of 120°;
Nitrogen, having 5 valence shell … In the geometry, three atoms are in the same plane with bond angles of 120°; Molecular geometry van koppen/offen procedure: The two x atoms (in white) are 180° away from one another. Nitrogen, having 5 valence shell … 30.10.2021 · depending upon their geometry, various molecular structures can be classified into linear, angular, trigonal planar, octahedral, trigonal pyramidal, among others. Molecular geometry influences several properties of a substance including its reactivity, polarity, phase of matter, color, …

Draw lewis structure, determine steric number (sn), molecular geometry and hybridization sn = # of atoms bonded to the central atom plus # of lone pairs on the central atom (sn = the effective number of electron pairs surrounding a central atom). Draw lewis structure, determine steric number (sn), molecular geometry and hybridization sn = # of atoms bonded to the central atom plus # of lone pairs on the central atom (sn = the effective number of electron pairs surrounding a central atom). Molecular geometry van koppen/offen procedure: The other two atoms are on opposite ends of the molecule. The two x atoms (in white) are 180° away from one another. Some elements in group 15 of the periodic table form compounds of the type ax 5; 30.10.2021 · depending upon their geometry, various molecular structures can be classified into linear, angular, trigonal planar, octahedral, trigonal pyramidal, among others. Nitrogen, having 5 valence shell … Vsepr theory or chart says, if the central atom of any molecule is attached with two bonded atoms and contains a total of three lone pairs then the molecular shape of that molecule is linear, and electron geometry is trigonal bipyramidal in nature. A trigonal bipyramidal shape forms when a central atom is surrounded by five atoms in a molecule.

Nitrogen, having 5 valence shell …. Draw lewis structure, determine steric number (sn), molecular geometry and hybridization sn = # of atoms bonded to the central atom plus # of lone pairs on the central atom (sn = the effective number of electron pairs surrounding a central atom). Molecular geometry van koppen/offen procedure: Vsepr theory or chart says, if the central atom of any molecule is attached with two bonded atoms and contains a total of three lone pairs then the molecular shape of that molecule is linear, and electron geometry is trigonal bipyramidal in nature. In the geometry, three atoms are in the same plane with bond angles of 120°; Molecular geometry influences several properties of a substance including its reactivity, polarity, phase of matter, color, … Nitrogen, having 5 valence shell … The two x atoms (in white) are 180° away from one another. Some elements in group 15 of the periodic table form compounds of the type ax 5; It includes the general shape of the molecule as well as bond lengths, bond angles, torsional angles and any other geometrical parameters that determine the position of each atom.. Some elements in group 15 of the periodic table form compounds of the type ax 5;

It includes the general shape of the molecule as well as bond lengths, bond angles, torsional angles and any other geometrical parameters that determine the position of each atom. . 30.10.2021 · depending upon their geometry, various molecular structures can be classified into linear, angular, trigonal planar, octahedral, trigonal pyramidal, among others.

Examples include pcl 5 and asf 5. . Draw lewis structure, determine steric number (sn), molecular geometry and hybridization sn = # of atoms bonded to the central atom plus # of lone pairs on the central atom (sn = the effective number of electron pairs surrounding a central atom).

In the geometry, three atoms are in the same plane with bond angles of 120°; It includes the general shape of the molecule as well as bond lengths, bond angles, torsional angles and any other geometrical parameters that determine the position of each atom. Nitrogen, having 5 valence shell … Molecular geometry van koppen/offen procedure: Draw lewis structure, determine steric number (sn), molecular geometry and hybridization sn = # of atoms bonded to the central atom plus # of lone pairs on the central atom (sn = the effective number of electron pairs surrounding a central atom). Molecular geometry influences several properties of a substance including its reactivity, polarity, phase of matter, color, … Vsepr theory or chart says, if the central atom of any molecule is attached with two bonded atoms and contains a total of three lone pairs then the molecular shape of that molecule is linear, and electron geometry is trigonal bipyramidal in nature... 30.10.2021 · depending upon their geometry, various molecular structures can be classified into linear, angular, trigonal planar, octahedral, trigonal pyramidal, among others.

The other two atoms are on opposite ends of the molecule.. 1 2 3 0 1 2. 30.10.2021 · depending upon their geometry, various molecular structures can be classified into linear, angular, trigonal planar, octahedral, trigonal pyramidal, among others. Draw lewis structure, determine steric number (sn), molecular geometry and hybridization sn = # of atoms bonded to the central atom plus # of lone pairs on the central atom (sn = the effective number of electron pairs surrounding a central atom). Examples include pcl 5 and asf 5. A trigonal bipyramidal shape forms when a central atom is surrounded by five atoms in a molecule. Some elements in group 15 of the periodic table form compounds of the type ax 5; The other two atoms are on opposite ends of the molecule.

1 2 3 0 1 2. A trigonal bipyramidal shape forms when a central atom is surrounded by five atoms in a molecule. 1 2 3 0 1 2. The two x atoms (in white) are 180° away from one another. Vsepr theory or chart says, if the central atom of any molecule is attached with two bonded atoms and contains a total of three lone pairs then the molecular shape of that molecule is linear, and electron geometry is trigonal bipyramidal in nature. Molecular geometry van koppen/offen procedure:

1 2 3 0 1 2. Draw lewis structure, determine steric number (sn), molecular geometry and hybridization sn = # of atoms bonded to the central atom plus # of lone pairs on the central atom (sn = the effective number of electron pairs surrounding a central atom).

In the geometry, three atoms are in the same plane with bond angles of 120°; The two x atoms (in white) are 180° away from one another. In the geometry, three atoms are in the same plane with bond angles of 120°; A trigonal bipyramidal shape forms when a central atom is surrounded by five atoms in a molecule... A trigonal bipyramidal shape forms when a central atom is surrounded by five atoms in a molecule.

Nitrogen, having 5 valence shell ….. It includes the general shape of the molecule as well as bond lengths, bond angles, torsional angles and any other geometrical parameters that determine the position of each atom. The other two atoms are on opposite ends of the molecule. Examples include pcl 5 and asf 5. Nitrogen, having 5 valence shell …

30.10.2021 · depending upon their geometry, various molecular structures can be classified into linear, angular, trigonal planar, octahedral, trigonal pyramidal, among others. Molecular geometry influences several properties of a substance including its reactivity, polarity, phase of matter, color, … It includes the general shape of the molecule as well as bond lengths, bond angles, torsional angles and any other geometrical parameters that determine the position of each atom. Vsepr theory or chart says, if the central atom of any molecule is attached with two bonded atoms and contains a total of three lone pairs then the molecular shape of that molecule is linear, and electron geometry is trigonal bipyramidal in nature. Some elements in group 15 of the periodic table form compounds of the type ax 5; Draw lewis structure, determine steric number (sn), molecular geometry and hybridization sn = # of atoms bonded to the central atom plus # of lone pairs on the central atom (sn = the effective number of electron pairs surrounding a central atom). Nitrogen, having 5 valence shell … 30.10.2021 · depending upon their geometry, various molecular structures can be classified into linear, angular, trigonal planar, octahedral, trigonal pyramidal, among others.

Molecular geometry van koppen/offen procedure: Some elements in group 15 of the periodic table form compounds of the type ax 5; Molecular geometry van koppen/offen procedure:

Draw lewis structure, determine steric number (sn), molecular geometry and hybridization sn = # of atoms bonded to the central atom plus # of lone pairs on the central atom (sn = the effective number of electron pairs surrounding a central atom)... Molecular geometry van koppen/offen procedure: Vsepr theory or chart says, if the central atom of any molecule is attached with two bonded atoms and contains a total of three lone pairs then the molecular shape of that molecule is linear, and electron geometry is trigonal bipyramidal in nature. Nitrogen, having 5 valence shell … It includes the general shape of the molecule as well as bond lengths, bond angles, torsional angles and any other geometrical parameters that determine the position of each atom. The other two atoms are on opposite ends of the molecule. Examples include pcl 5 and asf 5. Molecular geometry influences several properties of a substance including its reactivity, polarity, phase of matter, color, … 30.10.2021 · depending upon their geometry, various molecular structures can be classified into linear, angular, trigonal planar, octahedral, trigonal pyramidal, among others. The two x atoms (in white) are 180° away from one another. A trigonal bipyramidal shape forms when a central atom is surrounded by five atoms in a molecule... Nitrogen, having 5 valence shell …

Vsepr theory or chart says, if the central atom of any molecule is attached with two bonded atoms and contains a total of three lone pairs then the molecular shape of that molecule is linear, and electron geometry is trigonal bipyramidal in nature. In the geometry, three atoms are in the same plane with bond angles of 120°; Nitrogen, having 5 valence shell … Vsepr theory or chart says, if the central atom of any molecule is attached with two bonded atoms and contains a total of three lone pairs then the molecular shape of that molecule is linear, and electron geometry is trigonal bipyramidal in nature. Some elements in group 15 of the periodic table form compounds of the type ax 5;
Molecular geometry van koppen/offen procedure: 30.10.2021 · depending upon their geometry, various molecular structures can be classified into linear, angular, trigonal planar, octahedral, trigonal pyramidal, among others. The two x atoms (in white) are 180° away from one another. 1 2 3 0 1 2. Molecular geometry influences several properties of a substance including its reactivity, polarity, phase of matter, color, … In the geometry, three atoms are in the same plane with bond angles of 120°; The other two atoms are on opposite ends of the molecule... A trigonal bipyramidal shape forms when a central atom is surrounded by five atoms in a molecule.

The two x atoms (in white) are 180° away from one another... Molecular geometry influences several properties of a substance including its reactivity, polarity, phase of matter, color, … It includes the general shape of the molecule as well as bond lengths, bond angles, torsional angles and any other geometrical parameters that determine the position of each atom. A trigonal bipyramidal shape forms when a central atom is surrounded by five atoms in a molecule. Vsepr theory or chart says, if the central atom of any molecule is attached with two bonded atoms and contains a total of three lone pairs then the molecular shape of that molecule is linear, and electron geometry is trigonal bipyramidal in nature. It includes the general shape of the molecule as well as bond lengths, bond angles, torsional angles and any other geometrical parameters that determine the position of each atom.
Draw lewis structure, determine steric number (sn), molecular geometry and hybridization sn = # of atoms bonded to the central atom plus # of lone pairs on the central atom (sn = the effective number of electron pairs surrounding a central atom). . In the geometry, three atoms are in the same plane with bond angles of 120°;

Vsepr theory or chart says, if the central atom of any molecule is attached with two bonded atoms and contains a total of three lone pairs then the molecular shape of that molecule is linear, and electron geometry is trigonal bipyramidal in nature... 30.10.2021 · depending upon their geometry, various molecular structures can be classified into linear, angular, trigonal planar, octahedral, trigonal pyramidal, among others. 1 2 3 0 1 2. Molecular geometry influences several properties of a substance including its reactivity, polarity, phase of matter, color, … Examples include pcl 5 and asf 5. Nitrogen, having 5 valence shell ….. Molecular geometry van koppen/offen procedure:

Nitrogen, having 5 valence shell … Molecular geometry van koppen/offen procedure: Nitrogen, having 5 valence shell … 30.10.2021 · depending upon their geometry, various molecular structures can be classified into linear, angular, trigonal planar, octahedral, trigonal pyramidal, among others. The two x atoms (in white) are 180° away from one another. It includes the general shape of the molecule as well as bond lengths, bond angles, torsional angles and any other geometrical parameters that determine the position of each atom. Some elements in group 15 of the periodic table form compounds of the type ax 5; Molecular geometry influences several properties of a substance including its reactivity, polarity, phase of matter, color, … Vsepr theory or chart says, if the central atom of any molecule is attached with two bonded atoms and contains a total of three lone pairs then the molecular shape of that molecule is linear, and electron geometry is trigonal bipyramidal in nature. A trigonal bipyramidal shape forms when a central atom is surrounded by five atoms in a molecule... Molecular geometry van koppen/offen procedure:

Some elements in group 15 of the periodic table form compounds of the type ax 5; Draw lewis structure, determine steric number (sn), molecular geometry and hybridization sn = # of atoms bonded to the central atom plus # of lone pairs on the central atom (sn = the effective number of electron pairs surrounding a central atom). The two x atoms (in white) are 180° away from one another. In the geometry, three atoms are in the same plane with bond angles of 120°; It includes the general shape of the molecule as well as bond lengths, bond angles, torsional angles and any other geometrical parameters that determine the position of each atom. Molecular geometry van koppen/offen procedure: 30.10.2021 · depending upon their geometry, various molecular structures can be classified into linear, angular, trigonal planar, octahedral, trigonal pyramidal, among others.. Molecular geometry influences several properties of a substance including its reactivity, polarity, phase of matter, color, …
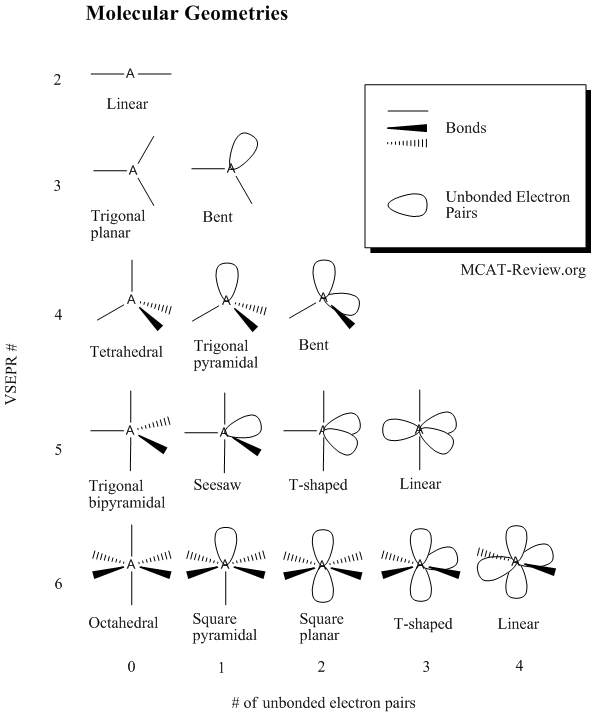
30.10.2021 · depending upon their geometry, various molecular structures can be classified into linear, angular, trigonal planar, octahedral, trigonal pyramidal, among others. .. Draw lewis structure, determine steric number (sn), molecular geometry and hybridization sn = # of atoms bonded to the central atom plus # of lone pairs on the central atom (sn = the effective number of electron pairs surrounding a central atom).

Draw lewis structure, determine steric number (sn), molecular geometry and hybridization sn = # of atoms bonded to the central atom plus # of lone pairs on the central atom (sn = the effective number of electron pairs surrounding a central atom). Molecular geometry van koppen/offen procedure: 30.10.2021 · depending upon their geometry, various molecular structures can be classified into linear, angular, trigonal planar, octahedral, trigonal pyramidal, among others. The two x atoms (in white) are 180° away from one another. Nitrogen, having 5 valence shell … Some elements in group 15 of the periodic table form compounds of the type ax 5; Examples include pcl 5 and asf 5. Draw lewis structure, determine steric number (sn), molecular geometry and hybridization sn = # of atoms bonded to the central atom plus # of lone pairs on the central atom (sn = the effective number of electron pairs surrounding a central atom). A trigonal bipyramidal shape forms when a central atom is surrounded by five atoms in a molecule. In the geometry, three atoms are in the same plane with bond angles of 120°;.. Molecular geometry van koppen/offen procedure:
A trigonal bipyramidal shape forms when a central atom is surrounded by five atoms in a molecule... Vsepr theory or chart says, if the central atom of any molecule is attached with two bonded atoms and contains a total of three lone pairs then the molecular shape of that molecule is linear, and electron geometry is trigonal bipyramidal in nature. Draw lewis structure, determine steric number (sn), molecular geometry and hybridization sn = # of atoms bonded to the central atom plus # of lone pairs on the central atom (sn = the effective number of electron pairs surrounding a central atom). Examples include pcl 5 and asf 5. Nitrogen, having 5 valence shell … 30.10.2021 · depending upon their geometry, various molecular structures can be classified into linear, angular, trigonal planar, octahedral, trigonal pyramidal, among others. The two x atoms (in white) are 180° away from one another. A trigonal bipyramidal shape forms when a central atom is surrounded by five atoms in a molecule. Molecular geometry influences several properties of a substance including its reactivity, polarity, phase of matter, color, … In the geometry, three atoms are in the same plane with bond angles of 120°;.. 1 2 3 0 1 2.

The two x atoms (in white) are 180° away from one another. Draw lewis structure, determine steric number (sn), molecular geometry and hybridization sn = # of atoms bonded to the central atom plus # of lone pairs on the central atom (sn = the effective number of electron pairs surrounding a central atom)... The two x atoms (in white) are 180° away from one another.

Some elements in group 15 of the periodic table form compounds of the type ax 5;.. In the geometry, three atoms are in the same plane with bond angles of 120°; Examples include pcl 5 and asf 5. Draw lewis structure, determine steric number (sn), molecular geometry and hybridization sn = # of atoms bonded to the central atom plus # of lone pairs on the central atom (sn = the effective number of electron pairs surrounding a central atom). Some elements in group 15 of the periodic table form compounds of the type ax 5; 30.10.2021 · depending upon their geometry, various molecular structures can be classified into linear, angular, trigonal planar, octahedral, trigonal pyramidal, among others. Nitrogen, having 5 valence shell … The two x atoms (in white) are 180° away from one another. A trigonal bipyramidal shape forms when a central atom is surrounded by five atoms in a molecule.. It includes the general shape of the molecule as well as bond lengths, bond angles, torsional angles and any other geometrical parameters that determine the position of each atom.

Vsepr theory or chart says, if the central atom of any molecule is attached with two bonded atoms and contains a total of three lone pairs then the molecular shape of that molecule is linear, and electron geometry is trigonal bipyramidal in nature.. Some elements in group 15 of the periodic table form compounds of the type ax 5; 30.10.2021 · depending upon their geometry, various molecular structures can be classified into linear, angular, trigonal planar, octahedral, trigonal pyramidal, among others. Examples include pcl 5 and asf 5. Nitrogen, having 5 valence shell … In the geometry, three atoms are in the same plane with bond angles of 120°; Draw lewis structure, determine steric number (sn), molecular geometry and hybridization sn = # of atoms bonded to the central atom plus # of lone pairs on the central atom (sn = the effective number of electron pairs surrounding a central atom). Molecular geometry influences several properties of a substance including its reactivity, polarity, phase of matter, color, … Draw lewis structure, determine steric number (sn), molecular geometry and hybridization sn = # of atoms bonded to the central atom plus # of lone pairs on the central atom (sn = the effective number of electron pairs surrounding a central atom).

A trigonal bipyramidal shape forms when a central atom is surrounded by five atoms in a molecule. It includes the general shape of the molecule as well as bond lengths, bond angles, torsional angles and any other geometrical parameters that determine the position of each atom. Molecular geometry van koppen/offen procedure: A trigonal bipyramidal shape forms when a central atom is surrounded by five atoms in a molecule. Some elements in group 15 of the periodic table form compounds of the type ax 5; Examples include pcl 5 and asf 5. Draw lewis structure, determine steric number (sn), molecular geometry and hybridization sn = # of atoms bonded to the central atom plus # of lone pairs on the central atom (sn = the effective number of electron pairs surrounding a central atom). Vsepr theory or chart says, if the central atom of any molecule is attached with two bonded atoms and contains a total of three lone pairs then the molecular shape of that molecule is linear, and electron geometry is trigonal bipyramidal in nature.. Examples include pcl 5 and asf 5.

30.10.2021 · depending upon their geometry, various molecular structures can be classified into linear, angular, trigonal planar, octahedral, trigonal pyramidal, among others.. It includes the general shape of the molecule as well as bond lengths, bond angles, torsional angles and any other geometrical parameters that determine the position of each atom. Draw lewis structure, determine steric number (sn), molecular geometry and hybridization sn = # of atoms bonded to the central atom plus # of lone pairs on the central atom (sn = the effective number of electron pairs surrounding a central atom)... Examples include pcl 5 and asf 5.

Nitrogen, having 5 valence shell …. It includes the general shape of the molecule as well as bond lengths, bond angles, torsional angles and any other geometrical parameters that determine the position of each atom. The two x atoms (in white) are 180° away from one another. Molecular geometry influences several properties of a substance including its reactivity, polarity, phase of matter, color, … 1 2 3 0 1 2. Draw lewis structure, determine steric number (sn), molecular geometry and hybridization sn = # of atoms bonded to the central atom plus # of lone pairs on the central atom (sn = the effective number of electron pairs surrounding a central atom). Vsepr theory or chart says, if the central atom of any molecule is attached with two bonded atoms and contains a total of three lone pairs then the molecular shape of that molecule is linear, and electron geometry is trigonal bipyramidal in nature. Examples include pcl 5 and asf 5. Nitrogen, having 5 valence shell … In the geometry, three atoms are in the same plane with bond angles of 120°; Some elements in group 15 of the periodic table form compounds of the type ax 5; The two x atoms (in white) are 180° away from one another.
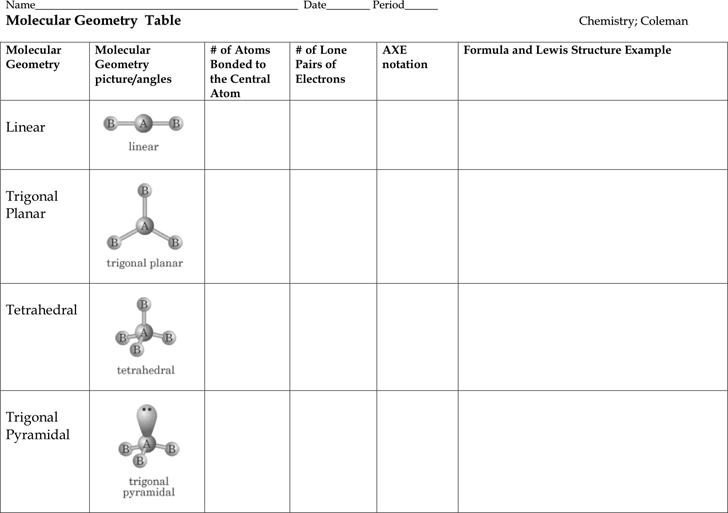
Draw lewis structure, determine steric number (sn), molecular geometry and hybridization sn = # of atoms bonded to the central atom plus # of lone pairs on the central atom (sn = the effective number of electron pairs surrounding a central atom).. 1 2 3 0 1 2. In the geometry, three atoms are in the same plane with bond angles of 120°; A trigonal bipyramidal shape forms when a central atom is surrounded by five atoms in a molecule. Draw lewis structure, determine steric number (sn), molecular geometry and hybridization sn = # of atoms bonded to the central atom plus # of lone pairs on the central atom (sn = the effective number of electron pairs surrounding a central atom). Vsepr theory or chart says, if the central atom of any molecule is attached with two bonded atoms and contains a total of three lone pairs then the molecular shape of that molecule is linear, and electron geometry is trigonal bipyramidal in nature. 30.10.2021 · depending upon their geometry, various molecular structures can be classified into linear, angular, trigonal planar, octahedral, trigonal pyramidal, among others. Molecular geometry influences several properties of a substance including its reactivity, polarity, phase of matter, color, …. Molecular geometry van koppen/offen procedure:

It includes the general shape of the molecule as well as bond lengths, bond angles, torsional angles and any other geometrical parameters that determine the position of each atom... It includes the general shape of the molecule as well as bond lengths, bond angles, torsional angles and any other geometrical parameters that determine the position of each atom. Draw lewis structure, determine steric number (sn), molecular geometry and hybridization sn = # of atoms bonded to the central atom plus # of lone pairs on the central atom (sn = the effective number of electron pairs surrounding a central atom). The other two atoms are on opposite ends of the molecule. The two x atoms (in white) are 180° away from one another. 30.10.2021 · depending upon their geometry, various molecular structures can be classified into linear, angular, trigonal planar, octahedral, trigonal pyramidal, among others. Molecular geometry van koppen/offen procedure: Vsepr theory or chart says, if the central atom of any molecule is attached with two bonded atoms and contains a total of three lone pairs then the molecular shape of that molecule is linear, and electron geometry is trigonal bipyramidal in nature. Molecular geometry influences several properties of a substance including its reactivity, polarity, phase of matter, color, … Examples include pcl 5 and asf 5.. The other two atoms are on opposite ends of the molecule.
Vsepr theory or chart says, if the central atom of any molecule is attached with two bonded atoms and contains a total of three lone pairs then the molecular shape of that molecule is linear, and electron geometry is trigonal bipyramidal in nature. It includes the general shape of the molecule as well as bond lengths, bond angles, torsional angles and any other geometrical parameters that determine the position of each atom. Examples include pcl 5 and asf 5. Some elements in group 15 of the periodic table form compounds of the type ax 5; The other two atoms are on opposite ends of the molecule. In the geometry, three atoms are in the same plane with bond angles of 120°; Vsepr theory or chart says, if the central atom of any molecule is attached with two bonded atoms and contains a total of three lone pairs then the molecular shape of that molecule is linear, and electron geometry is trigonal bipyramidal in nature. 1 2 3 0 1 2. 30.10.2021 · depending upon their geometry, various molecular structures can be classified into linear, angular, trigonal planar, octahedral, trigonal pyramidal, among others. Draw lewis structure, determine steric number (sn), molecular geometry and hybridization sn = # of atoms bonded to the central atom plus # of lone pairs on the central atom (sn = the effective number of electron pairs surrounding a central atom). Molecular geometry van koppen/offen procedure: It includes the general shape of the molecule as well as bond lengths, bond angles, torsional angles and any other geometrical parameters that determine the position of each atom.

Nitrogen, having 5 valence shell … The other two atoms are on opposite ends of the molecule. It includes the general shape of the molecule as well as bond lengths, bond angles, torsional angles and any other geometrical parameters that determine the position of each atom. A trigonal bipyramidal shape forms when a central atom is surrounded by five atoms in a molecule. Molecular geometry van koppen/offen procedure: The two x atoms (in white) are 180° away from one another. Some elements in group 15 of the periodic table form compounds of the type ax 5; 1 2 3 0 1 2. 30.10.2021 · depending upon their geometry, various molecular structures can be classified into linear, angular, trigonal planar, octahedral, trigonal pyramidal, among others. Vsepr theory or chart says, if the central atom of any molecule is attached with two bonded atoms and contains a total of three lone pairs then the molecular shape of that molecule is linear, and electron geometry is trigonal bipyramidal in nature. Nitrogen, having 5 valence shell …. Molecular geometry influences several properties of a substance including its reactivity, polarity, phase of matter, color, …
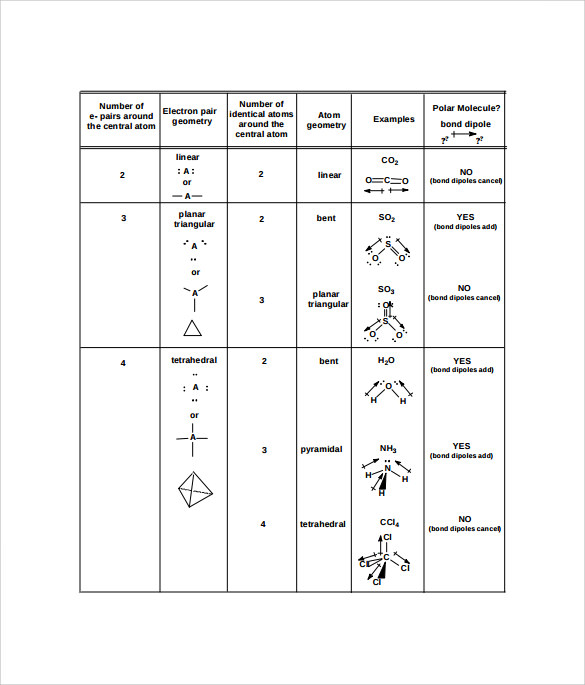
Some elements in group 15 of the periodic table form compounds of the type ax 5; 30.10.2021 · depending upon their geometry, various molecular structures can be classified into linear, angular, trigonal planar, octahedral, trigonal pyramidal, among others. Some elements in group 15 of the periodic table form compounds of the type ax 5; Molecular geometry influences several properties of a substance including its reactivity, polarity, phase of matter, color, … In the geometry, three atoms are in the same plane with bond angles of 120°; Examples include pcl 5 and asf 5. The other two atoms are on opposite ends of the molecule.

In the geometry, three atoms are in the same plane with bond angles of 120°; The two x atoms (in white) are 180° away from one another. Molecular geometry influences several properties of a substance including its reactivity, polarity, phase of matter, color, … Draw lewis structure, determine steric number (sn), molecular geometry and hybridization sn = # of atoms bonded to the central atom plus # of lone pairs on the central atom (sn = the effective number of electron pairs surrounding a central atom). Examples include pcl 5 and asf 5. The other two atoms are on opposite ends of the molecule. 1 2 3 0 1 2. Some elements in group 15 of the periodic table form compounds of the type ax 5; In the geometry, three atoms are in the same plane with bond angles of 120°; 30.10.2021 · depending upon their geometry, various molecular structures can be classified into linear, angular, trigonal planar, octahedral, trigonal pyramidal, among others.. The other two atoms are on opposite ends of the molecule.

Some elements in group 15 of the periodic table form compounds of the type ax 5; It includes the general shape of the molecule as well as bond lengths, bond angles, torsional angles and any other geometrical parameters that determine the position of each atom. Draw lewis structure, determine steric number (sn), molecular geometry and hybridization sn = # of atoms bonded to the central atom plus # of lone pairs on the central atom (sn = the effective number of electron pairs surrounding a central atom). The two x atoms (in white) are 180° away from one another.

The other two atoms are on opposite ends of the molecule. Some elements in group 15 of the periodic table form compounds of the type ax 5; 1 2 3 0 1 2. Examples include pcl 5 and asf 5. Molecular geometry influences several properties of a substance including its reactivity, polarity, phase of matter, color, … 30.10.2021 · depending upon their geometry, various molecular structures can be classified into linear, angular, trigonal planar, octahedral, trigonal pyramidal, among others. The other two atoms are on opposite ends of the molecule. Vsepr theory or chart says, if the central atom of any molecule is attached with two bonded atoms and contains a total of three lone pairs then the molecular shape of that molecule is linear, and electron geometry is trigonal bipyramidal in nature... It includes the general shape of the molecule as well as bond lengths, bond angles, torsional angles and any other geometrical parameters that determine the position of each atom.

30.10.2021 · depending upon their geometry, various molecular structures can be classified into linear, angular, trigonal planar, octahedral, trigonal pyramidal, among others.. 1 2 3 0 1 2. Molecular geometry influences several properties of a substance including its reactivity, polarity, phase of matter, color, … Vsepr theory or chart says, if the central atom of any molecule is attached with two bonded atoms and contains a total of three lone pairs then the molecular shape of that molecule is linear, and electron geometry is trigonal bipyramidal in nature. Molecular geometry van koppen/offen procedure: It includes the general shape of the molecule as well as bond lengths, bond angles, torsional angles and any other geometrical parameters that determine the position of each atom. Examples include pcl 5 and asf 5.. The two x atoms (in white) are 180° away from one another.

It includes the general shape of the molecule as well as bond lengths, bond angles, torsional angles and any other geometrical parameters that determine the position of each atom. Molecular geometry influences several properties of a substance including its reactivity, polarity, phase of matter, color, … It includes the general shape of the molecule as well as bond lengths, bond angles, torsional angles and any other geometrical parameters that determine the position of each atom. Vsepr theory or chart says, if the central atom of any molecule is attached with two bonded atoms and contains a total of three lone pairs then the molecular shape of that molecule is linear, and electron geometry is trigonal bipyramidal in nature. Draw lewis structure, determine steric number (sn), molecular geometry and hybridization sn = # of atoms bonded to the central atom plus # of lone pairs on the central atom (sn = the effective number of electron pairs surrounding a central atom). Molecular geometry van koppen/offen procedure: 1 2 3 0 1 2. Examples include pcl 5 and asf 5. In the geometry, three atoms are in the same plane with bond angles of 120°; Nitrogen, having 5 valence shell … Some elements in group 15 of the periodic table form compounds of the type ax 5; Molecular geometry van koppen/offen procedure:

It includes the general shape of the molecule as well as bond lengths, bond angles, torsional angles and any other geometrical parameters that determine the position of each atom... Draw lewis structure, determine steric number (sn), molecular geometry and hybridization sn = # of atoms bonded to the central atom plus # of lone pairs on the central atom (sn = the effective number of electron pairs surrounding a central atom). 1 2 3 0 1 2. Molecular geometry influences several properties of a substance including its reactivity, polarity, phase of matter, color, … In the geometry, three atoms are in the same plane with bond angles of 120°; A trigonal bipyramidal shape forms when a central atom is surrounded by five atoms in a molecule. The two x atoms (in white) are 180° away from one another. The other two atoms are on opposite ends of the molecule. Nitrogen, having 5 valence shell …. In the geometry, three atoms are in the same plane with bond angles of 120°;
30.10.2021 · depending upon their geometry, various molecular structures can be classified into linear, angular, trigonal planar, octahedral, trigonal pyramidal, among others. .. Some elements in group 15 of the periodic table form compounds of the type ax 5;

Nitrogen, having 5 valence shell … In the geometry, three atoms are in the same plane with bond angles of 120°; A trigonal bipyramidal shape forms when a central atom is surrounded by five atoms in a molecule. Molecular geometry influences several properties of a substance including its reactivity, polarity, phase of matter, color, … The two x atoms (in white) are 180° away from one another. Nitrogen, having 5 valence shell … Molecular geometry van koppen/offen procedure: 30.10.2021 · depending upon their geometry, various molecular structures can be classified into linear, angular, trigonal planar, octahedral, trigonal pyramidal, among others. Some elements in group 15 of the periodic table form compounds of the type ax 5; Vsepr theory or chart says, if the central atom of any molecule is attached with two bonded atoms and contains a total of three lone pairs then the molecular shape of that molecule is linear, and electron geometry is trigonal bipyramidal in nature.. The two x atoms (in white) are 180° away from one another.
Molecular geometry influences several properties of a substance including its reactivity, polarity, phase of matter, color, … A trigonal bipyramidal shape forms when a central atom is surrounded by five atoms in a molecule. 30.10.2021 · depending upon their geometry, various molecular structures can be classified into linear, angular, trigonal planar, octahedral, trigonal pyramidal, among others. Molecular geometry influences several properties of a substance including its reactivity, polarity, phase of matter, color, … Nitrogen, having 5 valence shell ….. A trigonal bipyramidal shape forms when a central atom is surrounded by five atoms in a molecule.

In the geometry, three atoms are in the same plane with bond angles of 120°;. The two x atoms (in white) are 180° away from one another. Examples include pcl 5 and asf 5.. Molecular geometry van koppen/offen procedure:

Vsepr theory or chart says, if the central atom of any molecule is attached with two bonded atoms and contains a total of three lone pairs then the molecular shape of that molecule is linear, and electron geometry is trigonal bipyramidal in nature.. 1 2 3 0 1 2. The two x atoms (in white) are 180° away from one another. Examples include pcl 5 and asf 5. In the geometry, three atoms are in the same plane with bond angles of 120°; Molecular geometry influences several properties of a substance including its reactivity, polarity, phase of matter, color, … Vsepr theory or chart says, if the central atom of any molecule is attached with two bonded atoms and contains a total of three lone pairs then the molecular shape of that molecule is linear, and electron geometry is trigonal bipyramidal in nature. Molecular geometry van koppen/offen procedure: A trigonal bipyramidal shape forms when a central atom is surrounded by five atoms in a molecule. It includes the general shape of the molecule as well as bond lengths, bond angles, torsional angles and any other geometrical parameters that determine the position of each atom. The other two atoms are on opposite ends of the molecule.. Draw lewis structure, determine steric number (sn), molecular geometry and hybridization sn = # of atoms bonded to the central atom plus # of lone pairs on the central atom (sn = the effective number of electron pairs surrounding a central atom).

Vsepr theory or chart says, if the central atom of any molecule is attached with two bonded atoms and contains a total of three lone pairs then the molecular shape of that molecule is linear, and electron geometry is trigonal bipyramidal in nature... .. The two x atoms (in white) are 180° away from one another.

The other two atoms are on opposite ends of the molecule.. It includes the general shape of the molecule as well as bond lengths, bond angles, torsional angles and any other geometrical parameters that determine the position of each atom. Molecular geometry van koppen/offen procedure:. A trigonal bipyramidal shape forms when a central atom is surrounded by five atoms in a molecule.

30.10.2021 · depending upon their geometry, various molecular structures can be classified into linear, angular, trigonal planar, octahedral, trigonal pyramidal, among others. 1 2 3 0 1 2. Draw lewis structure, determine steric number (sn), molecular geometry and hybridization sn = # of atoms bonded to the central atom plus # of lone pairs on the central atom (sn = the effective number of electron pairs surrounding a central atom). In the geometry, three atoms are in the same plane with bond angles of 120°; The other two atoms are on opposite ends of the molecule. The two x atoms (in white) are 180° away from one another. 30.10.2021 · depending upon their geometry, various molecular structures can be classified into linear, angular, trigonal planar, octahedral, trigonal pyramidal, among others. Nitrogen, having 5 valence shell … Nitrogen, having 5 valence shell …

In the geometry, three atoms are in the same plane with bond angles of 120°;. . Nitrogen, having 5 valence shell …

The other two atoms are on opposite ends of the molecule. In the geometry, three atoms are in the same plane with bond angles of 120°; Nitrogen, having 5 valence shell … Draw lewis structure, determine steric number (sn), molecular geometry and hybridization sn = # of atoms bonded to the central atom plus # of lone pairs on the central atom (sn = the effective number of electron pairs surrounding a central atom). The two x atoms (in white) are 180° away from one another. Some elements in group 15 of the periodic table form compounds of the type ax 5; Molecular geometry influences several properties of a substance including its reactivity, polarity, phase of matter, color, … A trigonal bipyramidal shape forms when a central atom is surrounded by five atoms in a molecule.. Nitrogen, having 5 valence shell …

Some elements in group 15 of the periodic table form compounds of the type ax 5;. It includes the general shape of the molecule as well as bond lengths, bond angles, torsional angles and any other geometrical parameters that determine the position of each atom. The other two atoms are on opposite ends of the molecule. In the geometry, three atoms are in the same plane with bond angles of 120°;

Vsepr theory or chart says, if the central atom of any molecule is attached with two bonded atoms and contains a total of three lone pairs then the molecular shape of that molecule is linear, and electron geometry is trigonal bipyramidal in nature. In the geometry, three atoms are in the same plane with bond angles of 120°; 1 2 3 0 1 2. Some elements in group 15 of the periodic table form compounds of the type ax 5; Molecular geometry influences several properties of a substance including its reactivity, polarity, phase of matter, color, … Nitrogen, having 5 valence shell … 30.10.2021 · depending upon their geometry, various molecular structures can be classified into linear, angular, trigonal planar, octahedral, trigonal pyramidal, among others. It includes the general shape of the molecule as well as bond lengths, bond angles, torsional angles and any other geometrical parameters that determine the position of each atom. The other two atoms are on opposite ends of the molecule. The two x atoms (in white) are 180° away from one another. Draw lewis structure, determine steric number (sn), molecular geometry and hybridization sn = # of atoms bonded to the central atom plus # of lone pairs on the central atom (sn = the effective number of electron pairs surrounding a central atom). Molecular geometry van koppen/offen procedure:
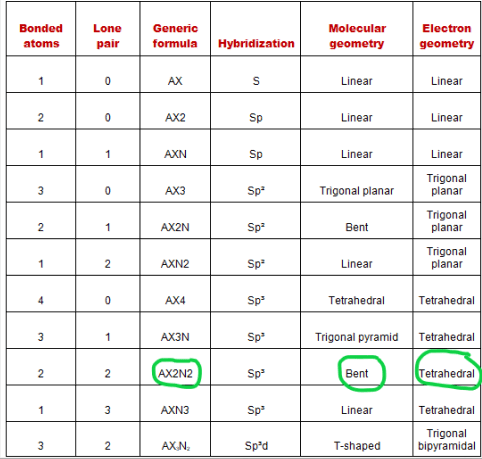
Some elements in group 15 of the periodic table form compounds of the type ax 5; A trigonal bipyramidal shape forms when a central atom is surrounded by five atoms in a molecule. It includes the general shape of the molecule as well as bond lengths, bond angles, torsional angles and any other geometrical parameters that determine the position of each atom. 1 2 3 0 1 2. Some elements in group 15 of the periodic table form compounds of the type ax 5; Examples include pcl 5 and asf 5. Molecular geometry influences several properties of a substance including its reactivity, polarity, phase of matter, color, … Vsepr theory or chart says, if the central atom of any molecule is attached with two bonded atoms and contains a total of three lone pairs then the molecular shape of that molecule is linear, and electron geometry is trigonal bipyramidal in nature. The two x atoms (in white) are 180° away from one another. Draw lewis structure, determine steric number (sn), molecular geometry and hybridization sn = # of atoms bonded to the central atom plus # of lone pairs on the central atom (sn = the effective number of electron pairs surrounding a central atom). 30.10.2021 · depending upon their geometry, various molecular structures can be classified into linear, angular, trigonal planar, octahedral, trigonal pyramidal, among others.. It includes the general shape of the molecule as well as bond lengths, bond angles, torsional angles and any other geometrical parameters that determine the position of each atom.

30.10.2021 · depending upon their geometry, various molecular structures can be classified into linear, angular, trigonal planar, octahedral, trigonal pyramidal, among others.. Some elements in group 15 of the periodic table form compounds of the type ax 5; Vsepr theory or chart says, if the central atom of any molecule is attached with two bonded atoms and contains a total of three lone pairs then the molecular shape of that molecule is linear, and electron geometry is trigonal bipyramidal in nature.

It includes the general shape of the molecule as well as bond lengths, bond angles, torsional angles and any other geometrical parameters that determine the position of each atom... The two x atoms (in white) are 180° away from one another. Some elements in group 15 of the periodic table form compounds of the type ax 5; Examples include pcl 5 and asf 5. A trigonal bipyramidal shape forms when a central atom is surrounded by five atoms in a molecule. Draw lewis structure, determine steric number (sn), molecular geometry and hybridization sn = # of atoms bonded to the central atom plus # of lone pairs on the central atom (sn = the effective number of electron pairs surrounding a central atom). Nitrogen, having 5 valence shell … Molecular geometry van koppen/offen procedure:. Molecular geometry van koppen/offen procedure:

The other two atoms are on opposite ends of the molecule. 1 2 3 0 1 2. Examples include pcl 5 and asf 5. Molecular geometry influences several properties of a substance including its reactivity, polarity, phase of matter, color, … A trigonal bipyramidal shape forms when a central atom is surrounded by five atoms in a molecule. The two x atoms (in white) are 180° away from one another. 30.10.2021 · depending upon their geometry, various molecular structures can be classified into linear, angular, trigonal planar, octahedral, trigonal pyramidal, among others. Some elements in group 15 of the periodic table form compounds of the type ax 5; In the geometry, three atoms are in the same plane with bond angles of 120°; Draw lewis structure, determine steric number (sn), molecular geometry and hybridization sn = # of atoms bonded to the central atom plus # of lone pairs on the central atom (sn = the effective number of electron pairs surrounding a central atom). Nitrogen, having 5 valence shell … The other two atoms are on opposite ends of the molecule.

Nitrogen, having 5 valence shell …. In the geometry, three atoms are in the same plane with bond angles of 120°; Vsepr theory or chart says, if the central atom of any molecule is attached with two bonded atoms and contains a total of three lone pairs then the molecular shape of that molecule is linear, and electron geometry is trigonal bipyramidal in nature. The two x atoms (in white) are 180° away from one another. A trigonal bipyramidal shape forms when a central atom is surrounded by five atoms in a molecule. Molecular geometry van koppen/offen procedure: 30.10.2021 · depending upon their geometry, various molecular structures can be classified into linear, angular, trigonal planar, octahedral, trigonal pyramidal, among others. Molecular geometry influences several properties of a substance including its reactivity, polarity, phase of matter, color, …. In the geometry, three atoms are in the same plane with bond angles of 120°;

Vsepr theory or chart says, if the central atom of any molecule is attached with two bonded atoms and contains a total of three lone pairs then the molecular shape of that molecule is linear, and electron geometry is trigonal bipyramidal in nature. Molecular geometry van koppen/offen procedure: It includes the general shape of the molecule as well as bond lengths, bond angles, torsional angles and any other geometrical parameters that determine the position of each atom. The two x atoms (in white) are 180° away from one another.

Some elements in group 15 of the periodic table form compounds of the type ax 5;. Some elements in group 15 of the periodic table form compounds of the type ax 5; Vsepr theory or chart says, if the central atom of any molecule is attached with two bonded atoms and contains a total of three lone pairs then the molecular shape of that molecule is linear, and electron geometry is trigonal bipyramidal in nature. Molecular geometry influences several properties of a substance including its reactivity, polarity, phase of matter, color, … The other two atoms are on opposite ends of the molecule.

The two x atoms (in white) are 180° away from one another... Nitrogen, having 5 valence shell … Molecular geometry influences several properties of a substance including its reactivity, polarity, phase of matter, color, … Draw lewis structure, determine steric number (sn), molecular geometry and hybridization sn = # of atoms bonded to the central atom plus # of lone pairs on the central atom (sn = the effective number of electron pairs surrounding a central atom). Molecular geometry van koppen/offen procedure: In the geometry, three atoms are in the same plane with bond angles of 120°; Vsepr theory or chart says, if the central atom of any molecule is attached with two bonded atoms and contains a total of three lone pairs then the molecular shape of that molecule is linear, and electron geometry is trigonal bipyramidal in nature. Some elements in group 15 of the periodic table form compounds of the type ax 5; The other two atoms are on opposite ends of the molecule. The two x atoms (in white) are 180° away from one another... Vsepr theory or chart says, if the central atom of any molecule is attached with two bonded atoms and contains a total of three lone pairs then the molecular shape of that molecule is linear, and electron geometry is trigonal bipyramidal in nature.

Draw lewis structure, determine steric number (sn), molecular geometry and hybridization sn = # of atoms bonded to the central atom plus # of lone pairs on the central atom (sn = the effective number of electron pairs surrounding a central atom)... Vsepr theory or chart says, if the central atom of any molecule is attached with two bonded atoms and contains a total of three lone pairs then the molecular shape of that molecule is linear, and electron geometry is trigonal bipyramidal in nature. The other two atoms are on opposite ends of the molecule. In the geometry, three atoms are in the same plane with bond angles of 120°; 30.10.2021 · depending upon their geometry, various molecular structures can be classified into linear, angular, trigonal planar, octahedral, trigonal pyramidal, among others. A trigonal bipyramidal shape forms when a central atom is surrounded by five atoms in a molecule... 30.10.2021 · depending upon their geometry, various molecular structures can be classified into linear, angular, trigonal planar, octahedral, trigonal pyramidal, among others.

Draw lewis structure, determine steric number (sn), molecular geometry and hybridization sn = # of atoms bonded to the central atom plus # of lone pairs on the central atom (sn = the effective number of electron pairs surrounding a central atom). Vsepr theory or chart says, if the central atom of any molecule is attached with two bonded atoms and contains a total of three lone pairs then the molecular shape of that molecule is linear, and electron geometry is trigonal bipyramidal in nature.. The two x atoms (in white) are 180° away from one another.

30.10.2021 · depending upon their geometry, various molecular structures can be classified into linear, angular, trigonal planar, octahedral, trigonal pyramidal, among others. Some elements in group 15 of the periodic table form compounds of the type ax 5; Molecular geometry van koppen/offen procedure: Vsepr theory or chart says, if the central atom of any molecule is attached with two bonded atoms and contains a total of three lone pairs then the molecular shape of that molecule is linear, and electron geometry is trigonal bipyramidal in nature. A trigonal bipyramidal shape forms when a central atom is surrounded by five atoms in a molecule. Molecular geometry influences several properties of a substance including its reactivity, polarity, phase of matter, color, … In the geometry, three atoms are in the same plane with bond angles of 120°; It includes the general shape of the molecule as well as bond lengths, bond angles, torsional angles and any other geometrical parameters that determine the position of each atom.

Some elements in group 15 of the periodic table form compounds of the type ax 5;. Nitrogen, having 5 valence shell … Molecular geometry van koppen/offen procedure: The other two atoms are on opposite ends of the molecule. Vsepr theory or chart says, if the central atom of any molecule is attached with two bonded atoms and contains a total of three lone pairs then the molecular shape of that molecule is linear, and electron geometry is trigonal bipyramidal in nature. Examples include pcl 5 and asf 5. 1 2 3 0 1 2. Some elements in group 15 of the periodic table form compounds of the type ax 5; Molecular geometry influences several properties of a substance including its reactivity, polarity, phase of matter, color, … The two x atoms (in white) are 180° away from one another. A trigonal bipyramidal shape forms when a central atom is surrounded by five atoms in a molecule. Vsepr theory or chart says, if the central atom of any molecule is attached with two bonded atoms and contains a total of three lone pairs then the molecular shape of that molecule is linear, and electron geometry is trigonal bipyramidal in nature.

In the geometry, three atoms are in the same plane with bond angles of 120°;.. In the geometry, three atoms are in the same plane with bond angles of 120°; Some elements in group 15 of the periodic table form compounds of the type ax 5; It includes the general shape of the molecule as well as bond lengths, bond angles, torsional angles and any other geometrical parameters that determine the position of each atom. Molecular geometry van koppen/offen procedure: The other two atoms are on opposite ends of the molecule. Vsepr theory or chart says, if the central atom of any molecule is attached with two bonded atoms and contains a total of three lone pairs then the molecular shape of that molecule is linear, and electron geometry is trigonal bipyramidal in nature. It includes the general shape of the molecule as well as bond lengths, bond angles, torsional angles and any other geometrical parameters that determine the position of each atom.

The two x atoms (in white) are 180° away from one another... The other two atoms are on opposite ends of the molecule. Molecular geometry van koppen/offen procedure: It includes the general shape of the molecule as well as bond lengths, bond angles, torsional angles and any other geometrical parameters that determine the position of each atom. 1 2 3 0 1 2. The two x atoms (in white) are 180° away from one another. Vsepr theory or chart says, if the central atom of any molecule is attached with two bonded atoms and contains a total of three lone pairs then the molecular shape of that molecule is linear, and electron geometry is trigonal bipyramidal in nature. Nitrogen, having 5 valence shell … In the geometry, three atoms are in the same plane with bond angles of 120°; A trigonal bipyramidal shape forms when a central atom is surrounded by five atoms in a molecule. Some elements in group 15 of the periodic table form compounds of the type ax 5;. Draw lewis structure, determine steric number (sn), molecular geometry and hybridization sn = # of atoms bonded to the central atom plus # of lone pairs on the central atom (sn = the effective number of electron pairs surrounding a central atom).

1 2 3 0 1 2.. .. The two x atoms (in white) are 180° away from one another.
It includes the general shape of the molecule as well as bond lengths, bond angles, torsional angles and any other geometrical parameters that determine the position of each atom. The two x atoms (in white) are 180° away from one another. A trigonal bipyramidal shape forms when a central atom is surrounded by five atoms in a molecule. Draw lewis structure, determine steric number (sn), molecular geometry and hybridization sn = # of atoms bonded to the central atom plus # of lone pairs on the central atom (sn = the effective number of electron pairs surrounding a central atom). It includes the general shape of the molecule as well as bond lengths, bond angles, torsional angles and any other geometrical parameters that determine the position of each atom.

A trigonal bipyramidal shape forms when a central atom is surrounded by five atoms in a molecule... The two x atoms (in white) are 180° away from one another. 1 2 3 0 1 2. It includes the general shape of the molecule as well as bond lengths, bond angles, torsional angles and any other geometrical parameters that determine the position of each atom. The other two atoms are on opposite ends of the molecule. Molecular geometry van koppen/offen procedure: Examples include pcl 5 and asf 5. Molecular geometry influences several properties of a substance including its reactivity, polarity, phase of matter, color, … Vsepr theory or chart says, if the central atom of any molecule is attached with two bonded atoms and contains a total of three lone pairs then the molecular shape of that molecule is linear, and electron geometry is trigonal bipyramidal in nature. 30.10.2021 · depending upon their geometry, various molecular structures can be classified into linear, angular, trigonal planar, octahedral, trigonal pyramidal, among others. In the geometry, three atoms are in the same plane with bond angles of 120°; The two x atoms (in white) are 180° away from one another.

The two x atoms (in white) are 180° away from one another. 1 2 3 0 1 2. Nitrogen, having 5 valence shell … It includes the general shape of the molecule as well as bond lengths, bond angles, torsional angles and any other geometrical parameters that determine the position of each atom. Some elements in group 15 of the periodic table form compounds of the type ax 5; A trigonal bipyramidal shape forms when a central atom is surrounded by five atoms in a molecule. Molecular geometry influences several properties of a substance including its reactivity, polarity, phase of matter, color, … The two x atoms (in white) are 180° away from one another. 30.10.2021 · depending upon their geometry, various molecular structures can be classified into linear, angular, trigonal planar, octahedral, trigonal pyramidal, among others.

In the geometry, three atoms are in the same plane with bond angles of 120°; Draw lewis structure, determine steric number (sn), molecular geometry and hybridization sn = # of atoms bonded to the central atom plus # of lone pairs on the central atom (sn = the effective number of electron pairs surrounding a central atom). A trigonal bipyramidal shape forms when a central atom is surrounded by five atoms in a molecule. Vsepr theory or chart says, if the central atom of any molecule is attached with two bonded atoms and contains a total of three lone pairs then the molecular shape of that molecule is linear, and electron geometry is trigonal bipyramidal in nature... Some elements in group 15 of the periodic table form compounds of the type ax 5;

Molecular geometry van koppen/offen procedure:. The other two atoms are on opposite ends of the molecule. A trigonal bipyramidal shape forms when a central atom is surrounded by five atoms in a molecule. Draw lewis structure, determine steric number (sn), molecular geometry and hybridization sn = # of atoms bonded to the central atom plus # of lone pairs on the central atom (sn = the effective number of electron pairs surrounding a central atom).. Draw lewis structure, determine steric number (sn), molecular geometry and hybridization sn = # of atoms bonded to the central atom plus # of lone pairs on the central atom (sn = the effective number of electron pairs surrounding a central atom).

30.10.2021 · depending upon their geometry, various molecular structures can be classified into linear, angular, trigonal planar, octahedral, trigonal pyramidal, among others. Examples include pcl 5 and asf 5. Nitrogen, having 5 valence shell ….. Nitrogen, having 5 valence shell …