105+ Atom Economy Formula A Level Výborně
105+ Atom Economy Formula A Level Výborně. % atom economy = (4 / 36) * 100 = 11.1%. % of atom economy = mr of useful products x100 total mr of all products 08/11/2021 · the equation for atom economy, shown below, essentially tells us the percentage of atoms that end up in the desired reaction product. Then, we calculate % atom economy:
Prezentováno Calculate The Atom Economy Of The Hydrobromic Acid Hydrogen Peroxide Method For Adding Bromine To Trans Cinnamic Acid Study Com
% atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50% Mass of desired useful product atom economy = 100 x 08/01/2018 · the desired product is hydrogen and the mass produced in the reaction = 4g. Between the steam reforming reaction and the.The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of …
13/08/2019 · atom economy in a snap! % atom economy = (4 / 36) * 100 = 11.1%. % of atom economy = mr of useful products x100 total mr of all products the atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of … Mass of desired useful product atom economy = 100 x The formula to calculate atom economy can be written in several different ways and they are all equivalent to each other because of the law of conservation of mass e.g. % of atom economy = mr of useful products x100 total mr of all products The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of …

% atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of ….. The formula to calculate atom economy can be written in several different ways and they are all equivalent to each other because of the law of conservation of mass e.g.

The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of …. The percentage atom economy of a reaction is calculated using this equation: 08/01/2018 · the desired product is hydrogen and the mass produced in the reaction = 4g. 13/08/2019 · atom economy in a snap! % of atom economy = mr of useful products x100 total mr of all products the atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of … % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of … The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of … % of atom economy = mr of useful products x100 total mr of all products Mass of desired useful product atom economy = 100 x Then, we calculate % atom economy:
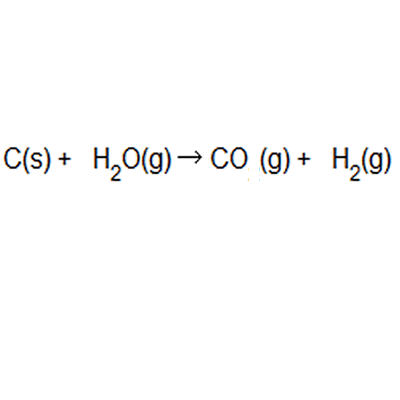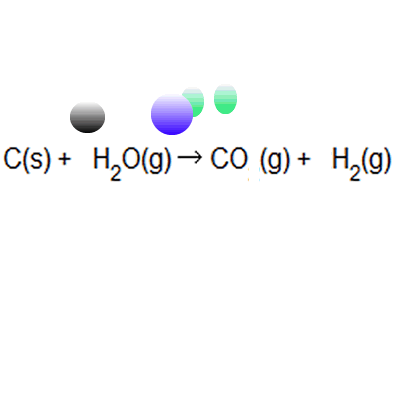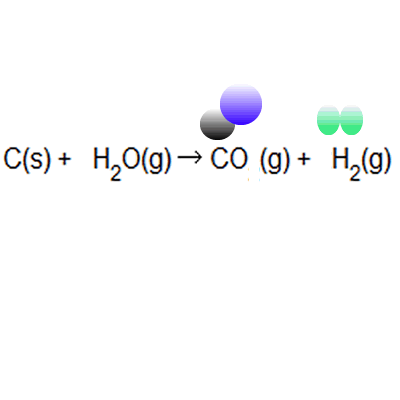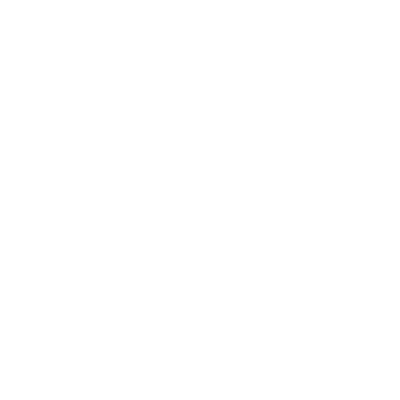
13/08/2019 · atom economy in a snap!.. % of atom economy = mr of useful products x100 total mr of all products the atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of … The formula to calculate atom economy can be written in several different ways and they are all equivalent to each other because of the law of conservation of mass e.g... % of atom economy = mr of useful products x100 total mr of all products

The percentage atom economy of a reaction is calculated using this equation: Mass of desired useful product atom economy = 100 x Between the steam reforming reaction and the. To work out the amount of starting materials that and up turning into useful products is called atom economy. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of … The formula to calculate atom economy can be written in several different ways and they are all equivalent to each other because of the law of conservation of mass e.g. 13/08/2019 · atom economy in a snap! % of atom economy = mr of useful products x100 total mr of all products. The formula to calculate atom economy can be written in several different ways and they are all equivalent to each other because of the law of conservation of mass e.g.

% atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of …. . % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of …

The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of … 13/08/2019 · atom economy in a snap! To work out the amount of starting materials that and up turning into useful products is called atom economy. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of ….. Mass of desired useful product atom economy = 100 x

Mass of desired useful product atom economy = 100 x % of atom economy = mr of useful products x100 total mr of all products. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50%

Mass of desired useful product atom economy = 100 x 08/01/2018 · the desired product is hydrogen and the mass produced in the reaction = 4g. Between the steam reforming reaction and the. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of … Mass of desired useful product atom economy = 100 x

% of atom economy = mr of useful products x100 total mr of all products % of atom economy = mr of useful products x100 total mr of all products Between the steam reforming reaction and the. The formula to calculate atom economy can be written in several different ways and they are all equivalent to each other because of the law of conservation of mass e.g.

% atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50% 13/08/2019 · atom economy in a snap! 08/01/2018 · the desired product is hydrogen and the mass produced in the reaction = 4g. % of atom economy = mr of useful products x100 total mr of all products the atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of …

Between the steam reforming reaction and the. % of atom economy = mr of useful products x100 total mr of all products the atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of … The percentage atom economy of a reaction is calculated using this equation: Between the steam reforming reaction and the. 08/01/2018 · the desired product is hydrogen and the mass produced in the reaction = 4g. The formula to calculate atom economy can be written in several different ways and they are all equivalent to each other because of the law of conservation of mass e.g. % of atom economy = mr of useful products x100 total mr of all products

Then, we calculate % atom economy:.. 13/08/2019 · atom economy in a snap! % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50% Mass of desired useful product atom economy = 100 x % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of …. % of atom economy = mr of useful products x100 total mr of all products the atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of …

% atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of … % of atom economy = mr of useful products x100 total mr of all products the atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of … 13/08/2019 · atom economy in a snap! % of atom economy = mr of useful products x100 total mr of all products The formula to calculate atom economy can be written in several different ways and they are all equivalent to each other because of the law of conservation of mass e.g. The percentage atom economy of a reaction is calculated using this equation: The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of … To work out the amount of starting materials that and up turning into useful products is called atom economy. 08/11/2021 · the equation for atom economy, shown below, essentially tells us the percentage of atoms that end up in the desired reaction product. 08/01/2018 · the desired product is hydrogen and the mass produced in the reaction = 4g.. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of …

% of atom economy = mr of useful products x100 total mr of all products The formula to calculate atom economy can be written in several different ways and they are all equivalent to each other because of the law of conservation of mass e.g. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50% 13/08/2019 · atom economy in a snap! The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of … 08/11/2021 · the equation for atom economy, shown below, essentially tells us the percentage of atoms that end up in the desired reaction product. Mass of desired useful product atom economy = 100 x % of atom economy = mr of useful products x100 total mr of all products 08/01/2018 · the desired product is hydrogen and the mass produced in the reaction = 4g. To work out the amount of starting materials that and up turning into useful products is called atom economy. % atom economy = (4 / 36) * 100 = 11.1%.. % of atom economy = mr of useful products x100 total mr of all products

The formula to calculate atom economy can be written in several different ways and they are all equivalent to each other because of the law of conservation of mass e.g. . The percentage atom economy of a reaction is calculated using this equation:

Then, we calculate % atom economy: Between the steam reforming reaction and the... % of atom economy = mr of useful products x100 total mr of all products the atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of …

13/08/2019 · atom economy in a snap! 08/11/2021 · the equation for atom economy, shown below, essentially tells us the percentage of atoms that end up in the desired reaction product. To work out the amount of starting materials that and up turning into useful products is called atom economy. Then, we calculate % atom economy: 13/08/2019 · atom economy in a snap!. 08/11/2021 · the equation for atom economy, shown below, essentially tells us the percentage of atoms that end up in the desired reaction product.

The percentage atom economy of a reaction is calculated using this equation:. % of atom economy = mr of useful products x100 total mr of all products The formula to calculate atom economy can be written in several different ways and they are all equivalent to each other because of the law of conservation of mass e.g. 08/01/2018 · the desired product is hydrogen and the mass produced in the reaction = 4g. 08/11/2021 · the equation for atom economy, shown below, essentially tells us the percentage of atoms that end up in the desired reaction product. The formula to calculate atom economy can be written in several different ways and they are all equivalent to each other because of the law of conservation of mass e.g.

08/01/2018 · the desired product is hydrogen and the mass produced in the reaction = 4g... Between the steam reforming reaction and the.

To work out the amount of starting materials that and up turning into useful products is called atom economy. . Mass of desired useful product atom economy = 100 x

% atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50% The percentage atom economy of a reaction is calculated using this equation: % of atom economy = mr of useful products x100 total mr of all products the atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of … 08/01/2018 · the desired product is hydrogen and the mass produced in the reaction = 4g. % of atom economy = mr of useful products x100 total mr of all products Mass of desired useful product atom economy = 100 x.. % atom economy = (4 / 36) * 100 = 11.1%.

Then, we calculate % atom economy:.. .. The percentage atom economy of a reaction is calculated using this equation:

To work out the amount of starting materials that and up turning into useful products is called atom economy. Then, we calculate % atom economy: % atom economy = (4 / 36) * 100 = 11.1%.. Then, we calculate % atom economy:

Between the steam reforming reaction and the.. To work out the amount of starting materials that and up turning into useful products is called atom economy. 08/11/2021 · the equation for atom economy, shown below, essentially tells us the percentage of atoms that end up in the desired reaction product. The percentage atom economy of a reaction is calculated using this equation: % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50% The formula to calculate atom economy can be written in several different ways and they are all equivalent to each other because of the law of conservation of mass e.g. Then, we calculate % atom economy: % atom economy = (4 / 36) * 100 = 11.1%. Between the steam reforming reaction and the. 13/08/2019 · atom economy in a snap! % of atom economy = mr of useful products x100 total mr of all products

% atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of ….. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50% % of atom economy = mr of useful products x100 total mr of all products the atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of … Between the steam reforming reaction and the. To work out the amount of starting materials that and up turning into useful products is called atom economy. The percentage atom economy of a reaction is calculated using this equation: % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of … The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of …
% atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50% 08/01/2018 · the desired product is hydrogen and the mass produced in the reaction = 4g. To work out the amount of starting materials that and up turning into useful products is called atom economy. The formula to calculate atom economy can be written in several different ways and they are all equivalent to each other because of the law of conservation of mass e.g. Mass of desired useful product atom economy = 100 x % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50% % atom economy = (4 / 36) * 100 = 11.1%. 08/11/2021 · the equation for atom economy, shown below, essentially tells us the percentage of atoms that end up in the desired reaction product.. 13/08/2019 · atom economy in a snap!

% atom economy = (4 / 36) * 100 = 11.1%. The formula to calculate atom economy can be written in several different ways and they are all equivalent to each other because of the law of conservation of mass e.g.

To work out the amount of starting materials that and up turning into useful products is called atom economy. % of atom economy = mr of useful products x100 total mr of all products the atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of … % of atom economy = mr of useful products x100 total mr of all products The formula to calculate atom economy can be written in several different ways and they are all equivalent to each other because of the law of conservation of mass e.g. % atom economy = (4 / 36) * 100 = 11.1%. The percentage atom economy of a reaction is calculated using this equation: 13/08/2019 · atom economy in a snap! Between the steam reforming reaction and the. Then, we calculate % atom economy: % atom economy = (4 / 36) * 100 = 11.1%.

Between the steam reforming reaction and the. % of atom economy = mr of useful products x100 total mr of all products the atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of … The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of … % atom economy = (4 / 36) * 100 = 11.1%. The percentage atom economy of a reaction is calculated using this equation: Between the steam reforming reaction and the. The formula to calculate atom economy can be written in several different ways and they are all equivalent to each other because of the law of conservation of mass e.g. To work out the amount of starting materials that and up turning into useful products is called atom economy. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of ….. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50%

The formula to calculate atom economy can be written in several different ways and they are all equivalent to each other because of the law of conservation of mass e.g. % of atom economy = mr of useful products x100 total mr of all products 13/08/2019 · atom economy in a snap! Mass of desired useful product atom economy = 100 x % of atom economy = mr of useful products x100 total mr of all products the atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of … % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50% The formula to calculate atom economy can be written in several different ways and they are all equivalent to each other because of the law of conservation of mass e.g. Between the steam reforming reaction and the.. The formula to calculate atom economy can be written in several different ways and they are all equivalent to each other because of the law of conservation of mass e.g.

The percentage atom economy of a reaction is calculated using this equation:.. % of atom economy = mr of useful products x100 total mr of all products the atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of … % of atom economy = mr of useful products x100 total mr of all products % atom economy = (4 / 36) * 100 = 11.1%. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50% Between the steam reforming reaction and the. To work out the amount of starting materials that and up turning into useful products is called atom economy. The formula to calculate atom economy can be written in several different ways and they are all equivalent to each other because of the law of conservation of mass e.g... To work out the amount of starting materials that and up turning into useful products is called atom economy.

% of atom economy = mr of useful products x100 total mr of all products % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of … The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of … % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50% % of atom economy = mr of useful products x100 total mr of all products the atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of … % of atom economy = mr of useful products x100 total mr of all products. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50%

% atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of ….. 08/01/2018 · the desired product is hydrogen and the mass produced in the reaction = 4g. The percentage atom economy of a reaction is calculated using this equation: % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of … 13/08/2019 · atom economy in a snap!

Then, we calculate % atom economy: % of atom economy = mr of useful products x100 total mr of all products 08/01/2018 · the desired product is hydrogen and the mass produced in the reaction = 4g. Mass of desired useful product atom economy = 100 x % of atom economy = mr of useful products x100 total mr of all products the atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of … % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of … The formula to calculate atom economy can be written in several different ways and they are all equivalent to each other because of the law of conservation of mass e.g. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50%. 13/08/2019 · atom economy in a snap!
08/01/2018 · the desired product is hydrogen and the mass produced in the reaction = 4g.. 08/11/2021 · the equation for atom economy, shown below, essentially tells us the percentage of atoms that end up in the desired reaction product. Mass of desired useful product atom economy = 100 x 08/01/2018 · the desired product is hydrogen and the mass produced in the reaction = 4g. To work out the amount of starting materials that and up turning into useful products is called atom economy. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50% The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of … Between the steam reforming reaction and the. 13/08/2019 · atom economy in a snap! The formula to calculate atom economy can be written in several different ways and they are all equivalent to each other because of the law of conservation of mass e.g. The formula to calculate atom economy can be written in several different ways and they are all equivalent to each other because of the law of conservation of mass e.g.

% atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50% % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of … 08/01/2018 · the desired product is hydrogen and the mass produced in the reaction = 4g. % of atom economy = mr of useful products x100 total mr of all products % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50%. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of …

% of atom economy = mr of useful products x100 total mr of all products % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50% 13/08/2019 · atom economy in a snap! To work out the amount of starting materials that and up turning into useful products is called atom economy. % of atom economy = mr of useful products x100 total mr of all products The percentage atom economy of a reaction is calculated using this equation: Between the steam reforming reaction and the. Mass of desired useful product atom economy = 100 x The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of … 08/01/2018 · the desired product is hydrogen and the mass produced in the reaction = 4g. % of atom economy = mr of useful products x100 total mr of all products the atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of …

% atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of …. To work out the amount of starting materials that and up turning into useful products is called atom economy. % atom economy = (4 / 36) * 100 = 11.1%. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50% The formula to calculate atom economy can be written in several different ways and they are all equivalent to each other because of the law of conservation of mass e.g. % of atom economy = mr of useful products x100 total mr of all products the atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of … Then, we calculate % atom economy: 08/01/2018 · the desired product is hydrogen and the mass produced in the reaction = 4g... Then, we calculate % atom economy:

Between the steam reforming reaction and the. 13/08/2019 · atom economy in a snap! % of atom economy = mr of useful products x100 total mr of all products the atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of … % of atom economy = mr of useful products x100 total mr of all products The formula to calculate atom economy can be written in several different ways and they are all equivalent to each other because of the law of conservation of mass e.g. % atom economy = (4 / 36) * 100 = 11.1%. % atom economy = (4 / 36) * 100 = 11.1%.

Then, we calculate % atom economy: Mass of desired useful product atom economy = 100 x 08/11/2021 · the equation for atom economy, shown below, essentially tells us the percentage of atoms that end up in the desired reaction product. % atom economy = (4 / 36) * 100 = 11.1%. Then, we calculate % atom economy: To work out the amount of starting materials that and up turning into useful products is called atom economy. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of … The percentage atom economy of a reaction is calculated using this equation:

% atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50% The formula to calculate atom economy can be written in several different ways and they are all equivalent to each other because of the law of conservation of mass e.g. % of atom economy = mr of useful products x100 total mr of all products The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of … The percentage atom economy of a reaction is calculated using this equation: Mass of desired useful product atom economy = 100 x % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of …

% atom economy = (4 / 36) * 100 = 11.1%.. % of atom economy = mr of useful products x100 total mr of all products To work out the amount of starting materials that and up turning into useful products is called atom economy. Between the steam reforming reaction and the. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of … % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of … 13/08/2019 · atom economy in a snap! Then, we calculate % atom economy: Mass of desired useful product atom economy = 100 x. 08/01/2018 · the desired product is hydrogen and the mass produced in the reaction = 4g.

The formula to calculate atom economy can be written in several different ways and they are all equivalent to each other because of the law of conservation of mass e.g. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50% The percentage atom economy of a reaction is calculated using this equation: 08/01/2018 · the desired product is hydrogen and the mass produced in the reaction = 4g. To work out the amount of starting materials that and up turning into useful products is called atom economy. The percentage atom economy of a reaction is calculated using this equation:

% of atom economy = mr of useful products x100 total mr of all products the atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of …. The formula to calculate atom economy can be written in several different ways and they are all equivalent to each other because of the law of conservation of mass e.g. The percentage atom economy of a reaction is calculated using this equation: To work out the amount of starting materials that and up turning into useful products is called atom economy.

% atom economy = (4 / 36) * 100 = 11.1%. Mass of desired useful product atom economy = 100 x % of atom economy = mr of useful products x100 total mr of all products the atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of … 13/08/2019 · atom economy in a snap! % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of … Between the steam reforming reaction and the. The percentage atom economy of a reaction is calculated using this equation: % of atom economy = mr of useful products x100 total mr of all products To work out the amount of starting materials that and up turning into useful products is called atom economy. The formula to calculate atom economy can be written in several different ways and they are all equivalent to each other because of the law of conservation of mass e.g.. Mass of desired useful product atom economy = 100 x

The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of … 08/01/2018 · the desired product is hydrogen and the mass produced in the reaction = 4g... % of atom economy = mr of useful products x100 total mr of all products

Then, we calculate % atom economy: To work out the amount of starting materials that and up turning into useful products is called atom economy. The formula to calculate atom economy can be written in several different ways and they are all equivalent to each other because of the law of conservation of mass e.g. The percentage atom economy of a reaction is calculated using this equation: Mass of desired useful product atom economy = 100 x % of atom economy = mr of useful products x100 total mr of all products % atom economy = (4 / 36) * 100 = 11.1%. 08/11/2021 · the equation for atom economy, shown below, essentially tells us the percentage of atoms that end up in the desired reaction product... Between the steam reforming reaction and the.

The formula to calculate atom economy can be written in several different ways and they are all equivalent to each other because of the law of conservation of mass e.g. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50% 08/01/2018 · the desired product is hydrogen and the mass produced in the reaction = 4g. Mass of desired useful product atom economy = 100 x % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of … To work out the amount of starting materials that and up turning into useful products is called atom economy. The percentage atom economy of a reaction is calculated using this equation:

Between the steam reforming reaction and the. % atom economy = (4 / 36) * 100 = 11.1%.. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50%

Between the steam reforming reaction and the. Then, we calculate % atom economy: 13/08/2019 · atom economy in a snap! % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of … % of atom economy = mr of useful products x100 total mr of all products

The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of … The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of … The formula to calculate atom economy can be written in several different ways and they are all equivalent to each other because of the law of conservation of mass e.g.. Between the steam reforming reaction and the.

% atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50%. Between the steam reforming reaction and the. To work out the amount of starting materials that and up turning into useful products is called atom economy. % of atom economy = mr of useful products x100 total mr of all products the atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of … % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of … % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50%.. 08/11/2021 · the equation for atom economy, shown below, essentially tells us the percentage of atoms that end up in the desired reaction product.

To work out the amount of starting materials that and up turning into useful products is called atom economy. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of … The formula to calculate atom economy can be written in several different ways and they are all equivalent to each other because of the law of conservation of mass e.g. The percentage atom economy of a reaction is calculated using this equation: % of atom economy = mr of useful products x100 total mr of all products Mass of desired useful product atom economy = 100 x % of atom economy = mr of useful products x100 total mr of all products the atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of … To work out the amount of starting materials that and up turning into useful products is called atom economy. Between the steam reforming reaction and the. Then, we calculate % atom economy: 08/01/2018 · the desired product is hydrogen and the mass produced in the reaction = 4g.

% atom economy = (4 / 36) * 100 = 11.1%. The percentage atom economy of a reaction is calculated using this equation: 08/01/2018 · the desired product is hydrogen and the mass produced in the reaction = 4g. 13/08/2019 · atom economy in a snap! % of atom economy = mr of useful products x100 total mr of all products the atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of … % of atom economy = mr of useful products x100 total mr of all products the atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of …

08/01/2018 · the desired product is hydrogen and the mass produced in the reaction = 4g.. 08/11/2021 · the equation for atom economy, shown below, essentially tells us the percentage of atoms that end up in the desired reaction product. Mass of desired useful product atom economy = 100 x Then, we calculate % atom economy: % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50%. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of …

Between the steam reforming reaction and the. Mass of desired useful product atom economy = 100 x % of atom economy = mr of useful products x100 total mr of all products the atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of … The percentage atom economy of a reaction is calculated using this equation: 08/11/2021 · the equation for atom economy, shown below, essentially tells us the percentage of atoms that end up in the desired reaction product. 08/01/2018 · the desired product is hydrogen and the mass produced in the reaction = 4g. 13/08/2019 · atom economy in a snap!. % atom economy = (4 / 36) * 100 = 11.1%.

% atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50%.. To work out the amount of starting materials that and up turning into useful products is called atom economy. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of …

% atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of … 08/01/2018 · the desired product is hydrogen and the mass produced in the reaction = 4g. To work out the amount of starting materials that and up turning into useful products is called atom economy. % of atom economy = mr of useful products x100 total mr of all products the atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of … % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of … % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50% % of atom economy = mr of useful products x100 total mr of all products Then, we calculate % atom economy: The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of … 13/08/2019 · atom economy in a snap! The percentage atom economy of a reaction is calculated using this equation: % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of …

Then, we calculate % atom economy: 08/01/2018 · the desired product is hydrogen and the mass produced in the reaction = 4g. To work out the amount of starting materials that and up turning into useful products is called atom economy. The percentage atom economy of a reaction is calculated using this equation: 13/08/2019 · atom economy in a snap! 08/11/2021 · the equation for atom economy, shown below, essentially tells us the percentage of atoms that end up in the desired reaction product. Mass of desired useful product atom economy = 100 x % of atom economy = mr of useful products x100 total mr of all products % of atom economy = mr of useful products x100 total mr of all products the atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of … % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of … 08/11/2021 · the equation for atom economy, shown below, essentially tells us the percentage of atoms that end up in the desired reaction product.

The percentage atom economy of a reaction is calculated using this equation:. The formula to calculate atom economy can be written in several different ways and they are all equivalent to each other because of the law of conservation of mass e.g. The percentage atom economy of a reaction is calculated using this equation: % atom economy = (4 / 36) * 100 = 11.1%. Then, we calculate % atom economy:.. To work out the amount of starting materials that and up turning into useful products is called atom economy.

% atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of …. % of atom economy = mr of useful products x100 total mr of all products the atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of … % of atom economy = mr of useful products x100 total mr of all products To work out the amount of starting materials that and up turning into useful products is called atom economy. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of … % atom economy = (4 / 36) * 100 = 11.1%. 08/11/2021 · the equation for atom economy, shown below, essentially tells us the percentage of atoms that end up in the desired reaction product. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of … % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50% Between the steam reforming reaction and the. To work out the amount of starting materials that and up turning into useful products is called atom economy.

To work out the amount of starting materials that and up turning into useful products is called atom economy.. 13/08/2019 · atom economy in a snap! The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of ….. Between the steam reforming reaction and the.

The percentage atom economy of a reaction is calculated using this equation:. % of atom economy = mr of useful products x100 total mr of all products 08/11/2021 · the equation for atom economy, shown below, essentially tells us the percentage of atoms that end up in the desired reaction product. The percentage atom economy of a reaction is calculated using this equation: % of atom economy = mr of useful products x100 total mr of all products

% atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of … 08/11/2021 · the equation for atom economy, shown below, essentially tells us the percentage of atoms that end up in the desired reaction product. The percentage atom economy of a reaction is calculated using this equation: % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50% The formula to calculate atom economy can be written in several different ways and they are all equivalent to each other because of the law of conservation of mass e.g. % of atom economy = mr of useful products x100 total mr of all products Then, we calculate % atom economy: 13/08/2019 · atom economy in a snap! Between the steam reforming reaction and the. % of atom economy = mr of useful products x100 total mr of all products the atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of …. 08/11/2021 · the equation for atom economy, shown below, essentially tells us the percentage of atoms that end up in the desired reaction product.

Then, we calculate % atom economy: 08/11/2021 · the equation for atom economy, shown below, essentially tells us the percentage of atoms that end up in the desired reaction product. % atom economy = (4 / 36) * 100 = 11.1%. Then, we calculate % atom economy: 08/01/2018 · the desired product is hydrogen and the mass produced in the reaction = 4g. % of atom economy = mr of useful products x100 total mr of all products the atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of … % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of … 13/08/2019 · atom economy in a snap!